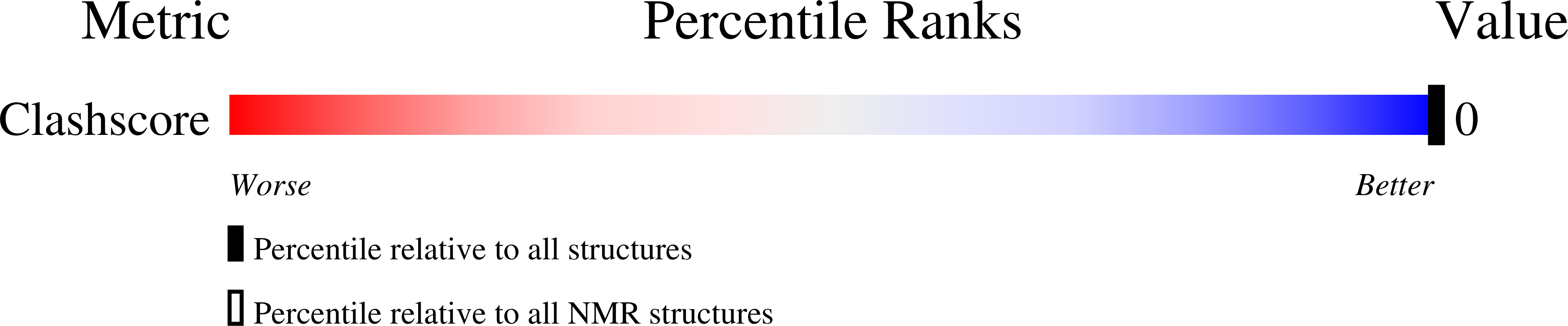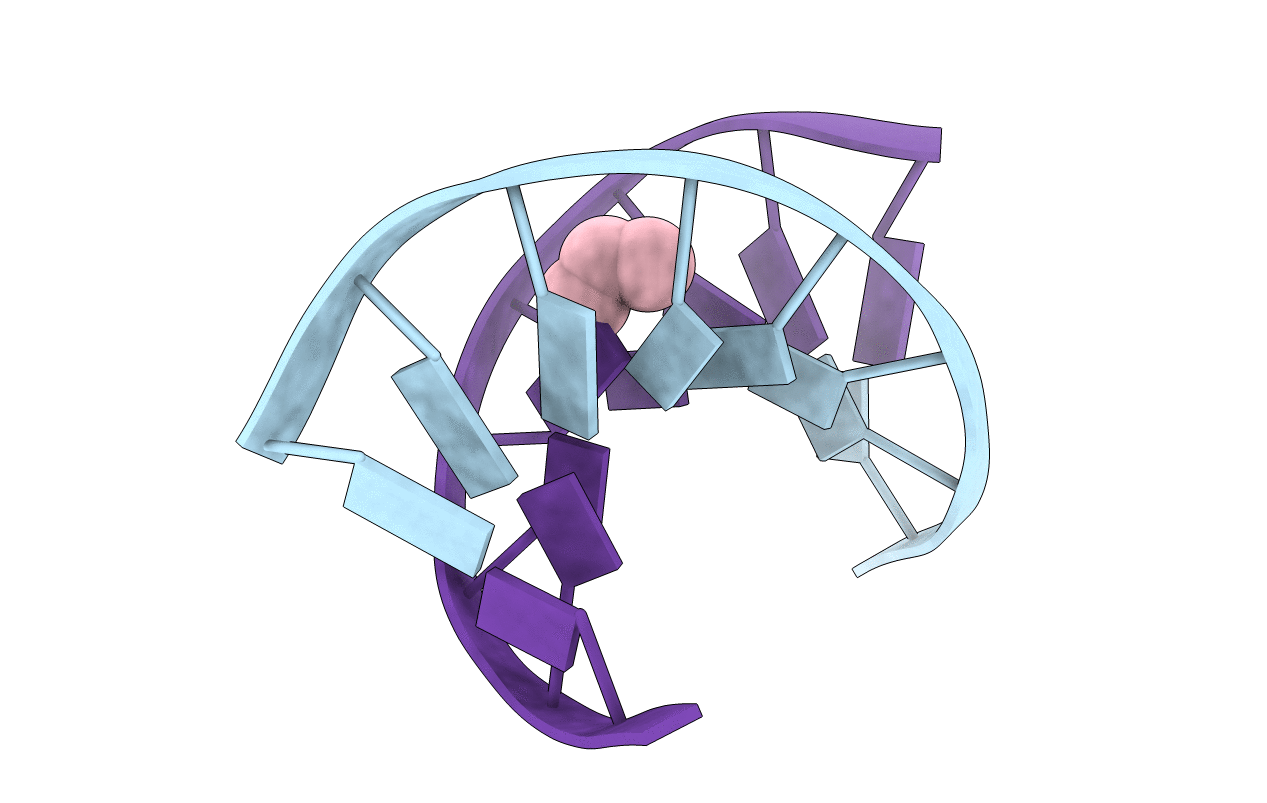
Deposition Date
2009-08-26
Release Date
2009-09-29
Last Version Date
2024-05-22
Entry Detail
PDB ID:
2KNL
Keywords:
Title:
Structure of the trimethylene N2-dG:N2-dG interstrand cross-link in the 5'-GpC-3' sequence context
Method Details:
Experimental Method:
Conformers Calculated:
20
Conformers Submitted:
1
Selection Criteria:
back calculated data agree with experimental NOESY spectrum