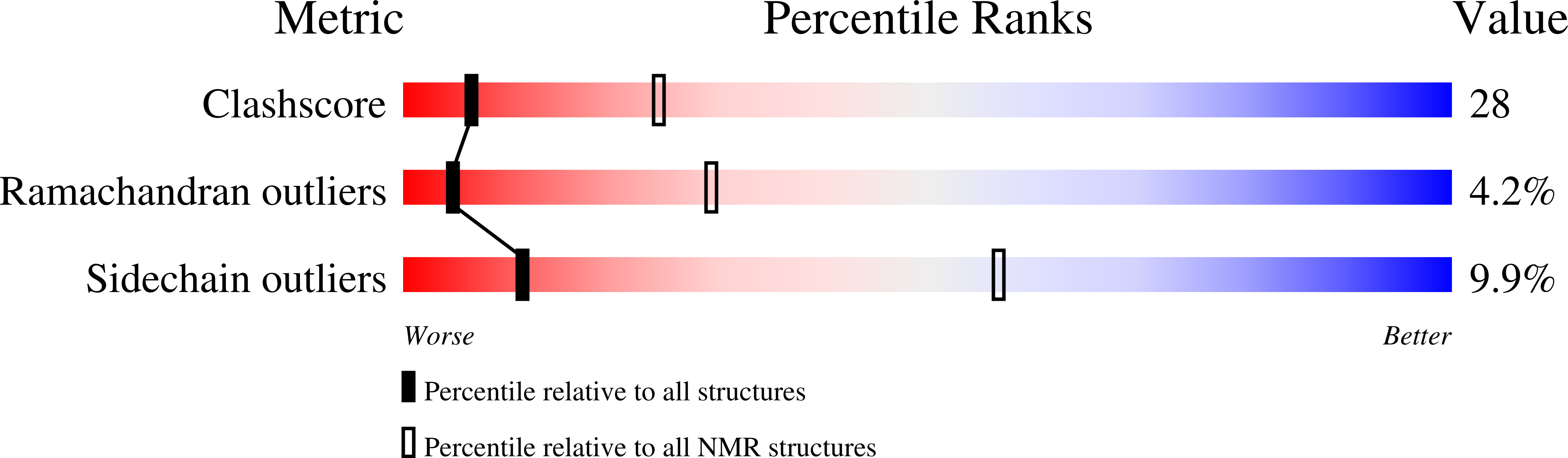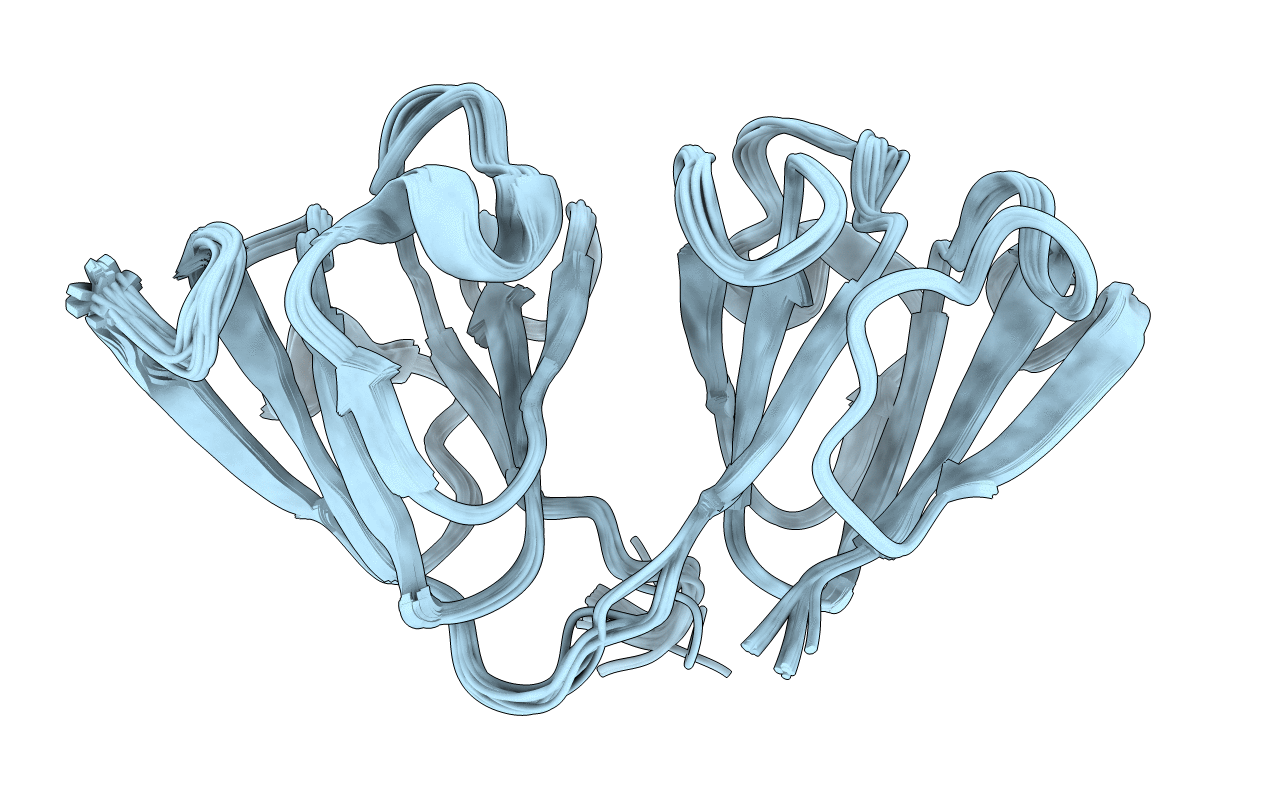
Deposition Date
2009-02-12
Release Date
2009-07-28
Last Version Date
2024-05-22
Entry Detail
PDB ID:
2KFB
Keywords:
Title:
The structure of the cataract causing P23T mutant of human gamma-D crystallin
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
Expression System(s):
Method Details:
Experimental Method:
Conformers Calculated:
100
Conformers Submitted:
20
Selection Criteria:
target function