Deposition Date
2005-07-08
Release Date
2005-09-20
Last Version Date
2024-02-14
Entry Detail
PDB ID:
2A8K
Keywords:
Title:
Structural and Mutational Studies of the Catalytic Domain of Colicin E5a tRNA-Specific Ribonuclease
Biological Source:
Source Organism(s):
Escherichia coli (Taxon ID: 562)
Expression System(s):
Method Details:
Experimental Method:
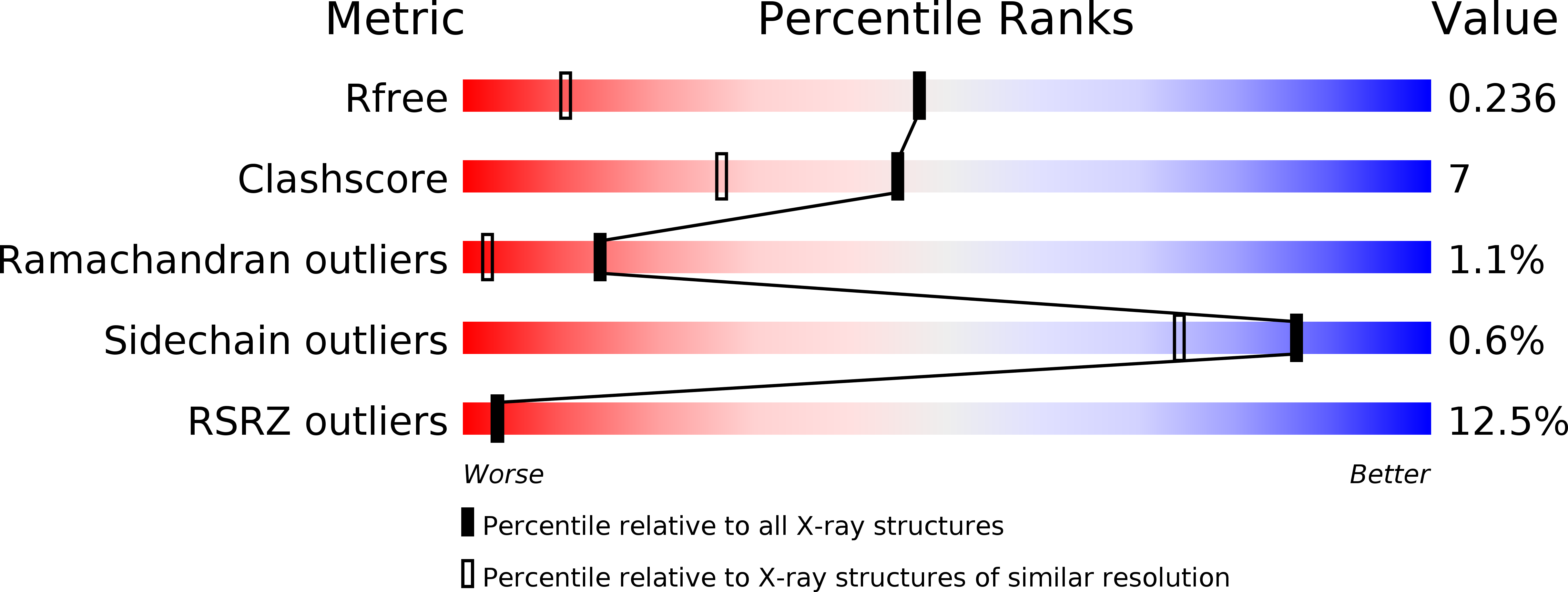
Resolution:
1.50 Å
R-Value Free:
0.23
R-Value Work:
0.21
Space Group:
P 31