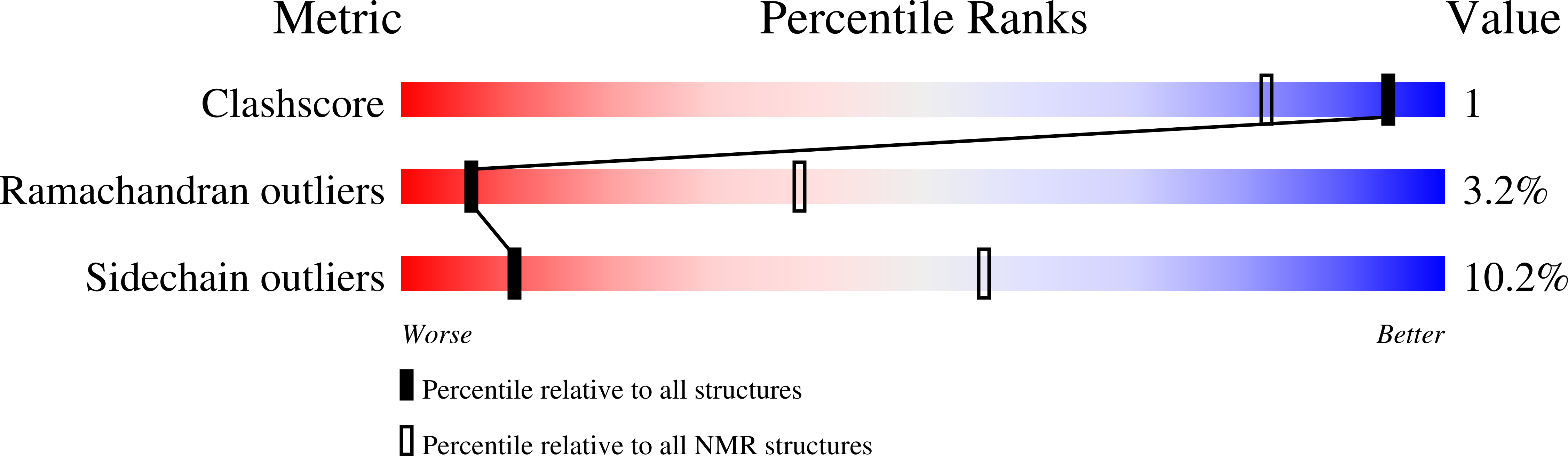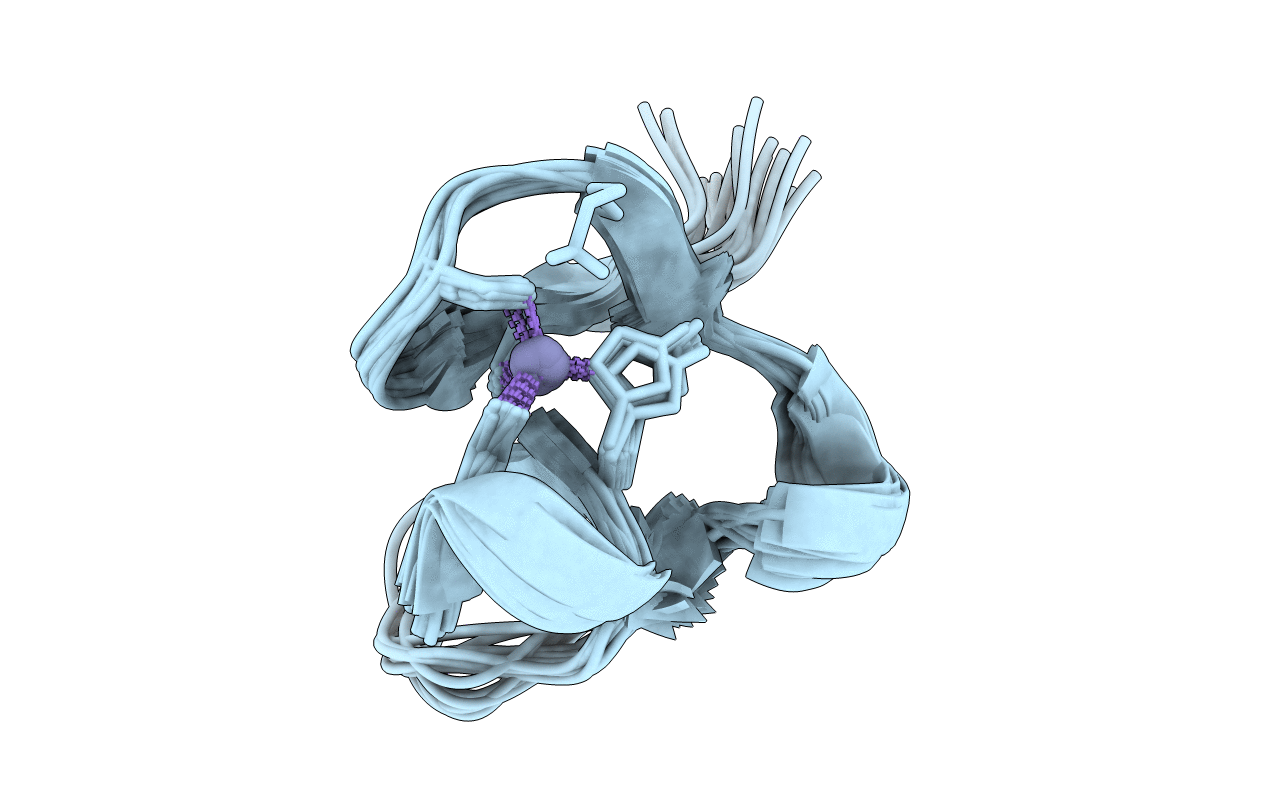
Deposition Date
1996-05-06
Release Date
1996-11-08
Last Version Date
2024-11-20
Entry Detail
PDB ID:
1ZFO
Keywords:
Title:
AMINO-TERMINAL LIM-DOMAIN PEPTIDE OF LASP-1, NMR
Biological Source:
Source Organism(s):
Sus scrofa (Taxon ID: 9823)
Method Details:
Experimental Method:
Conformers Submitted:
20