Deposition Date
2004-07-20
Release Date
2005-12-07
Last Version Date
2024-05-08
Entry Detail
PDB ID:
1W3X
Keywords:
Title:
Isopenicillin N synthase d-(L-a-aminoadipoyl)-(3R)-methyl-L-cysteine D-a-hydroxyisovaleryl ester complex (Oxygen exposed 5 minutes 20 bar)
Biological Source:
Source Organism:
Host Organism:
Method Details:
Experimental Method:
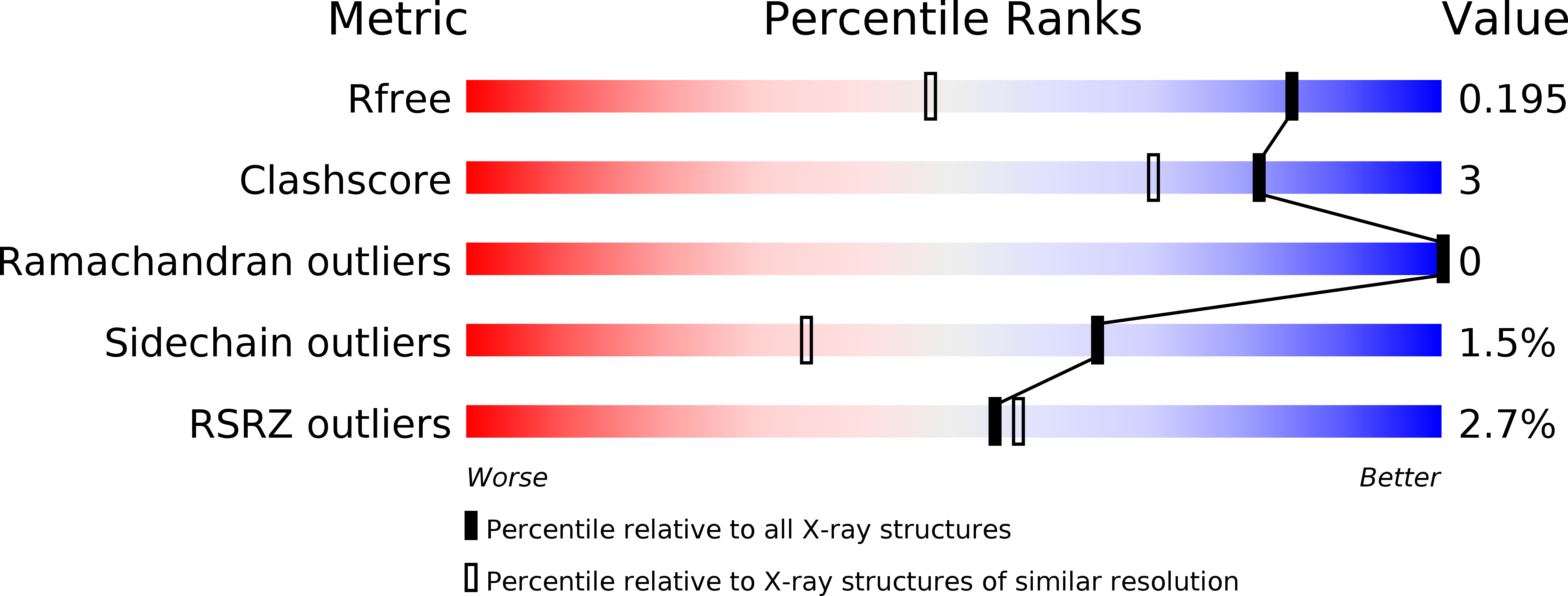
Resolution:
1.46 Å
R-Value Free:
0.18
R-Value Work:
0.16
R-Value Observed:
0.17
Space Group:
P 21 21 21