Deposition Date
1995-04-21
Release Date
1995-07-10
Last Version Date
2024-02-14
Entry Detail
PDB ID:
1VLZ
Keywords:
Title:
UNCOUPLED PHOSPHORYLATION AND ACTIVATION IN BACTERIAL CHEMOTAXIS: THE 2.1 ANGSTROM STRUCTURE OF A THREONINE TO ISOLEUCINE MUTANT AT POSITION 87 OF CHEY
Biological Source:
Source Organism(s):
Escherichia coli (Taxon ID: 562)
Method Details:
Experimental Method:
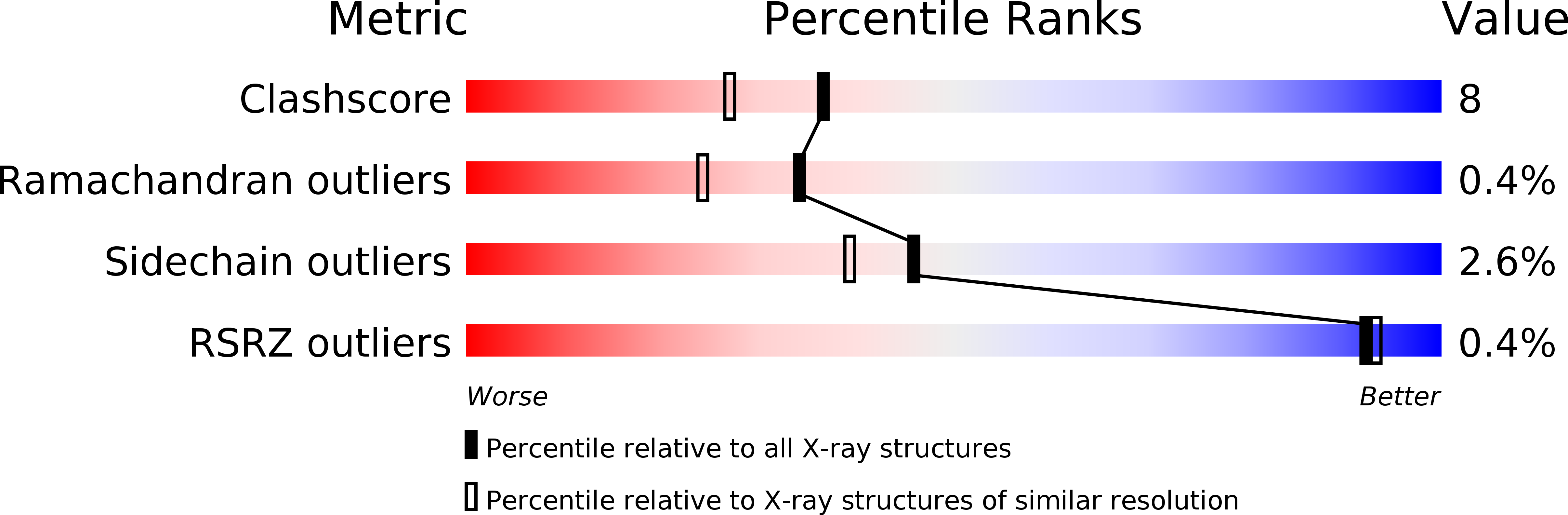
Resolution:
2.05 Å
R-Value Observed:
0.15
Space Group:
P 1 21 1