Abstact
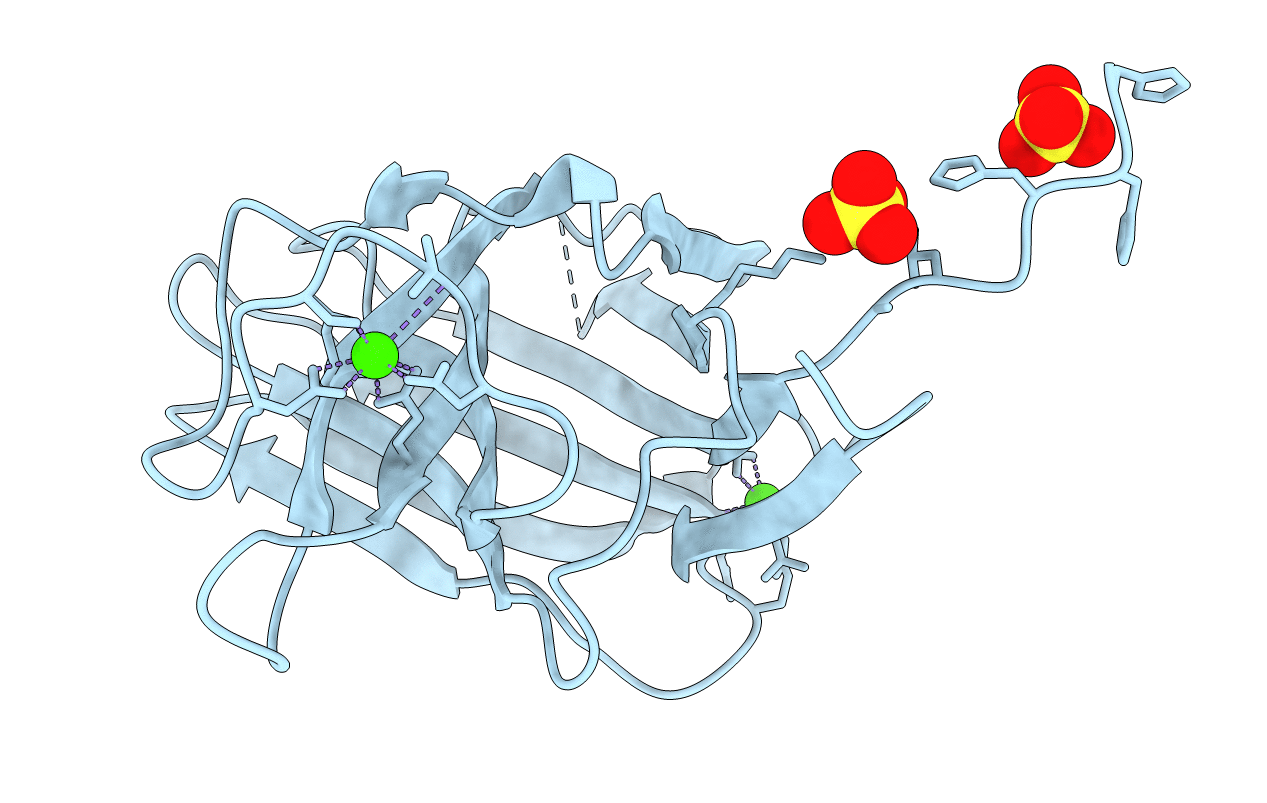
Modular glycoside hydrolases that attack recalcitrant polymers generally contain noncatalytic carbohydrate-binding modules (CBMs), which play a critical role in the action of these enzymes by localizing the appended catalytic domains onto the surface of insoluble polysaccharide substrates. Type B CBMs, which recognize single polysaccharide chains, display ligand specificities that are consistent with the substrates hydrolyzed by the associated catalytic domains. In enzymes that contain multiple catalytic domains with distinct substrate specificities, it is unclear how these different activities influence the evolution of the ligand recognition profile of the appended CBM. To address this issue, we have characterized the properties of a family 11 CBM (CtCBM11) in Clostridium thermocellum Lic26A-Cel5E, an enzyme that contains GH5 and GH26 catalytic domains that display beta-1,4- and beta-1,3-1,4-mixed linked endoglucanase activity, respectively. Here we show that CtCBM11 binds to both beta-1,4- and beta-1,3-1,4-mixed linked glucans, displaying K(a) values of 1.9 x 10(5), 4.4 x 10(4), and 2 x 10(3) m(-1) for Glc-beta1,4-Glc-beta1,4-Glc-beta1,3-Glc, Glc-beta1,4-Glc-beta1,4-Glc-beta1,4-Glc, and Glc-beta1,3-Glc-beta1,4-Glc-beta1,3-Glc, respectively, demonstrating that CBMs can display a preference for mixed linked glucans. To determine whether these ligands are accommodated in the same or diverse sites in CtCBM11, the crystal structure of the protein was solved to a resolution of 1.98 A. The protein displays a beta-sandwich with a concave side that forms a potential binding cleft. Site-directed mutagenesis revealed that Tyr(22), Tyr(53), and Tyr(129), located in the putative binding cleft, play a central role in the recognition of all the ligands recognized by the protein. We propose, therefore, that CtCBM11 contains a single ligand-binding site that displays affinity for both beta-1,4- and beta-1,3-1,4-mixed linked glucans.