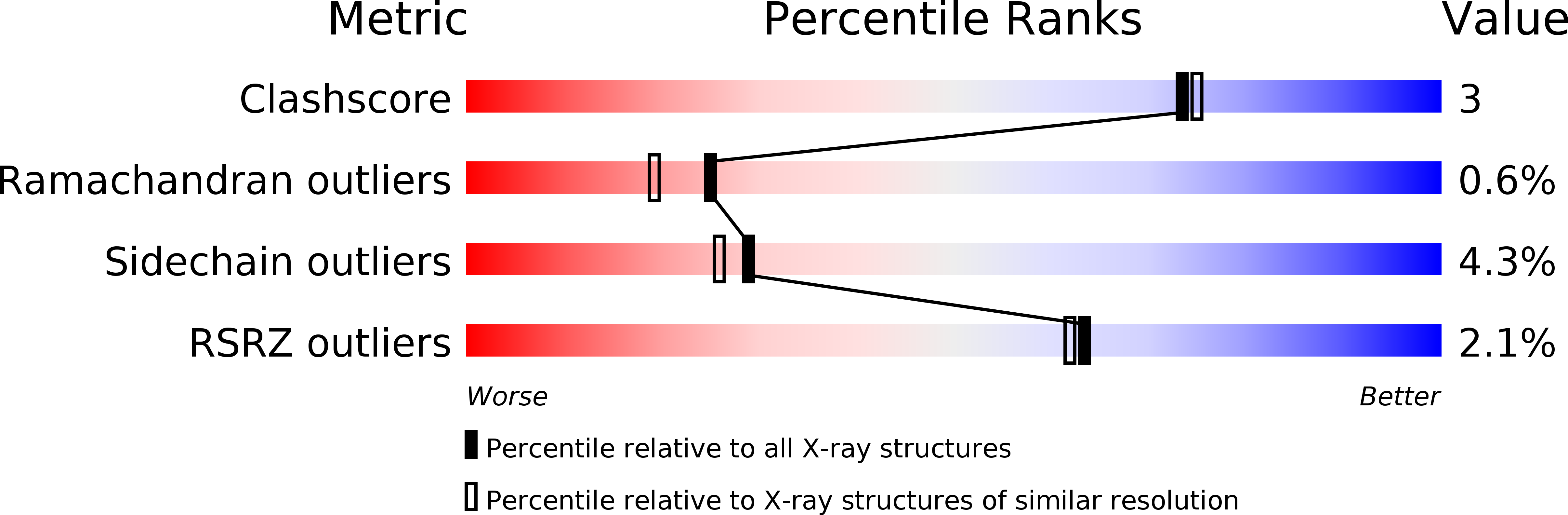Deposition Date
1997-02-04
Release Date
1998-09-16
Last Version Date
2024-04-03
Entry Detail
PDB ID:
1SZJ
Keywords:
Title:
STRUCTURE OF HOLO-GLYCERALDEHYDE-3-PHOSPHATE-DEHYDROGENASE FROM PALINURUS VERSICOLOR REFINED 2.0 ANGSTROM RESOLUTION
Biological Source:
Source Organism(s):
Palinurus versicolor (Taxon ID: 82835)
Method Details:
Experimental Method:
Resolution:
2.00 Å
R-Value Free:
0.22
R-Value Work:
0.16
R-Value Observed:
0.16
Space Group:
C 1 2 1