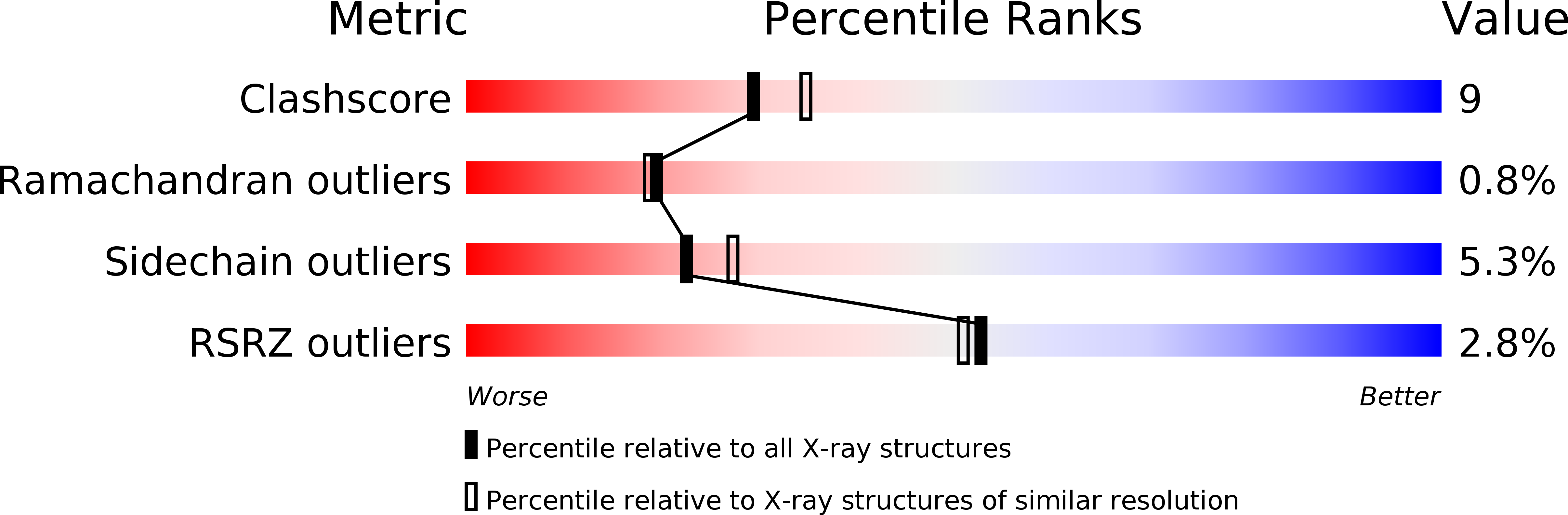
Deposition Date
2004-03-16
Release Date
2004-05-04
Last Version Date
2024-10-09
Entry Detail
PDB ID:
1SP4
Keywords:
Title:
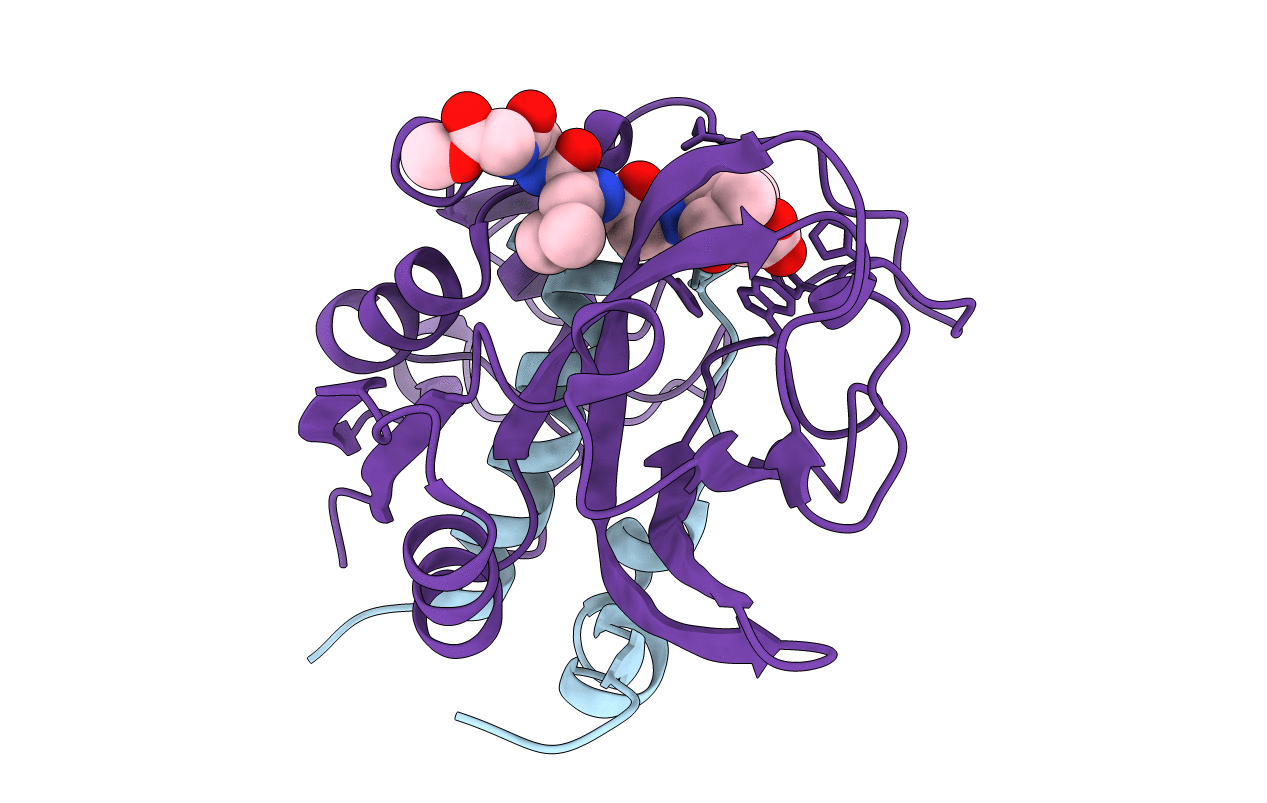
Crystal structure of NS-134 in complex with bovine cathepsin B: a two headed epoxysuccinyl inhibitor extends along the whole active site cleft
Biological Source:
Source Organism(s):
Bos taurus (Taxon ID: 9913)
Method Details: