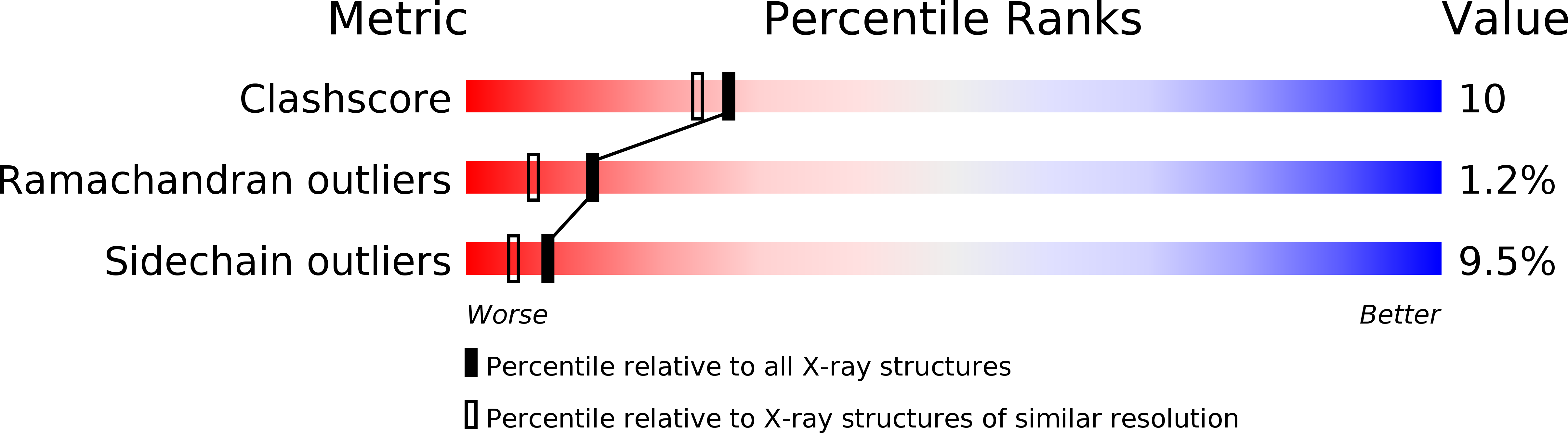Deposition Date
1992-03-11
Release Date
1994-01-31
Last Version Date
2025-03-26
Entry Detail
PDB ID:
1SMR
Keywords:
Title:
The 3-d structure of mouse submaxillary renin complexed with a decapeptide inhibitor ch-66 based on the 4-16 fragment of rat angiotensinogen
Biological Source:
Source Organism(s):
Mus musculus (Taxon ID: 10090)
Method Details:
Experimental Method:
Resolution:
2.00 Å
R-Value Observed:
0.18
Space Group:
P 1 21 1