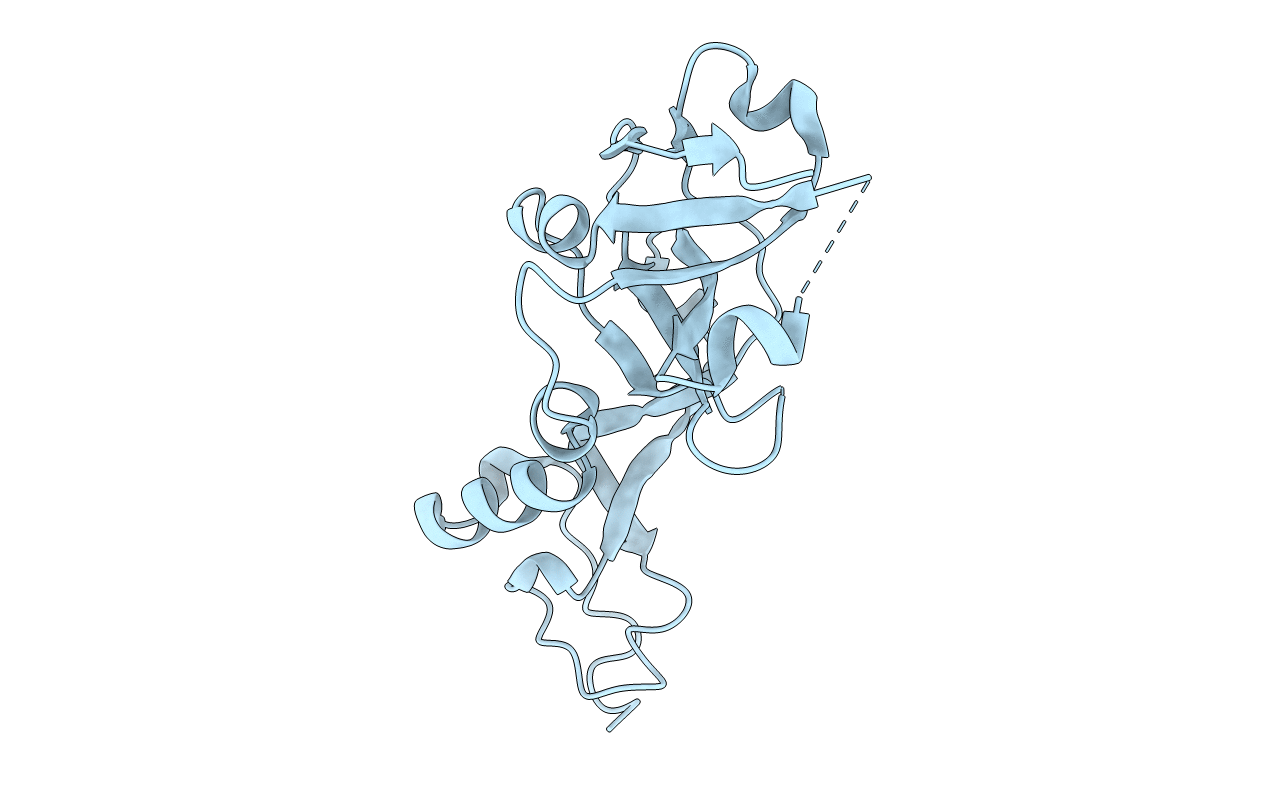
Deposition Date
2004-01-07
Release Date
2004-09-14
Last Version Date
2024-02-14
Entry Detail
PDB ID:
1S21
Keywords:
Title:
Crystal Structure of AvrPphF ORF2, A Type III Effector from P. syringae
Biological Source:
Source Organism(s):
Pseudomonas syringae pv. phaseolicola (Taxon ID: 319)
Expression System(s):
Method Details:
Experimental Method:
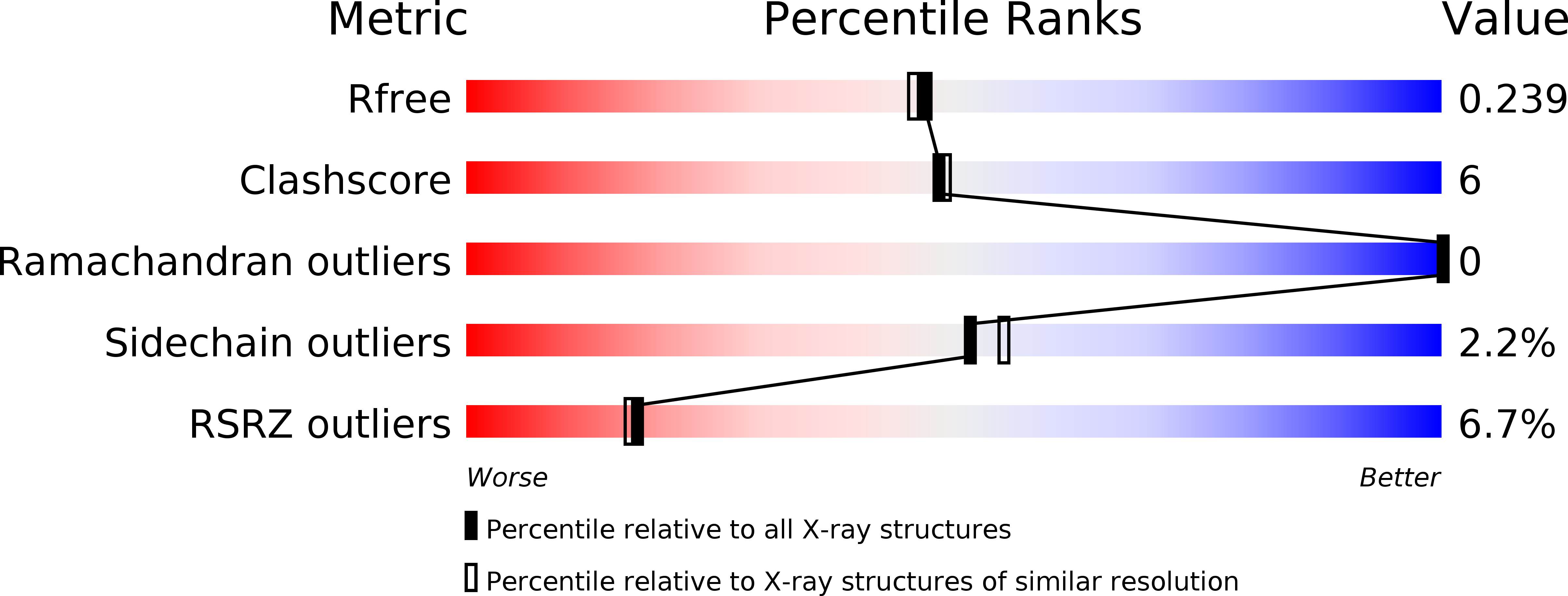
Resolution:
2.00 Å
R-Value Free:
0.24
R-Value Work:
0.22
R-Value Observed:
0.22
Space Group:
P 21 21 21