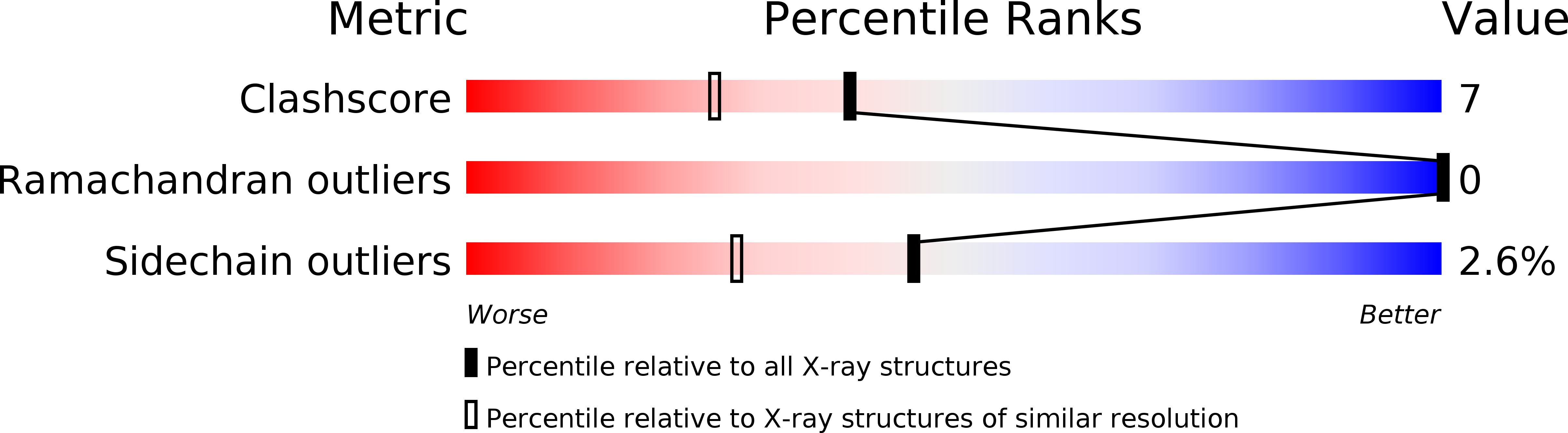Deposition Date
1999-06-22
Release Date
1999-07-02
Last Version Date
2024-02-14
Entry Detail
PDB ID:
1QSH
Keywords:
Title:
MAGNESIUM(II)-AND ZINC(II)-PROTOPORPHYRIN IX'S STABILIZE THE LOWEST OXYGEN AFFINITY STATE OF HUMAN HEMOGLOBIN EVEN MORE STRONGLY THAN DEOXYHEME
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
Method Details:
Experimental Method:
Resolution:
1.70 Å
R-Value Work:
0.17
R-Value Observed:
0.17
Space Group:
P 1 21 1