Deposition Date
2002-10-02
Release Date
2003-02-25
Last Version Date
2024-02-14
Entry Detail
PDB ID:
1MXI
Keywords:
Title:
Structure of YibK from Haemophilus influenzae (HI0766): a Methyltransferase with a Cofactor Bound at a Site Formed by a Knot
Biological Source:
Source Organism(s):
Haemophilus influenzae (Taxon ID: 727)
Expression System(s):
Method Details:
Experimental Method:
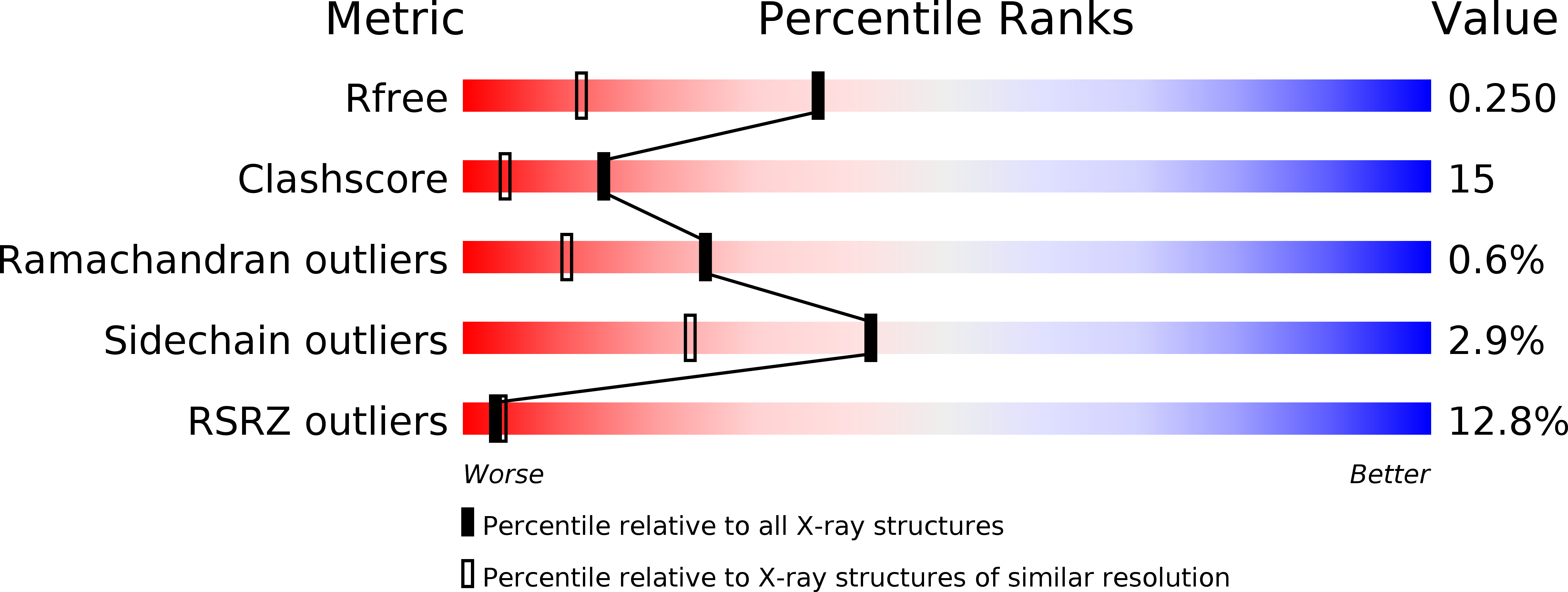
Resolution:
1.70 Å
R-Value Free:
0.25
R-Value Work:
0.19
Space Group:
P 41 21 2