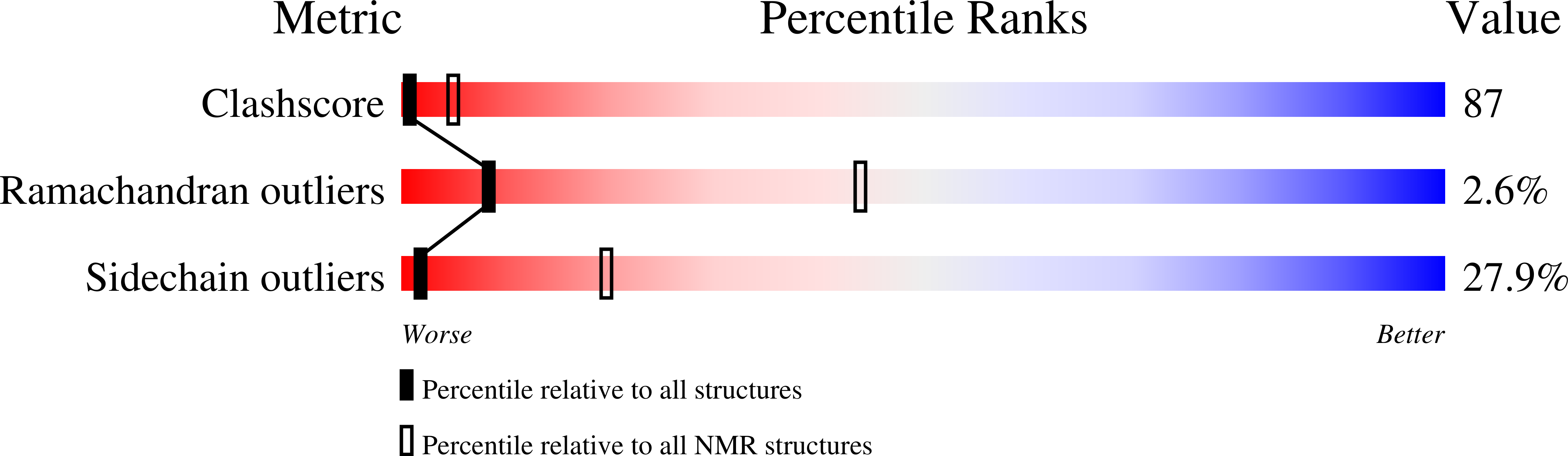Abstact
We report here the NMR structure and backbone dynamics of an exchangeable apolipoprotein, apoLp-III, from the insect Locusta migratoria. The NMR structure adopts an up-and-down elongated five-helix bundle, which is similar to the x-ray crystal structure of this protein. A short helix, helix 4', is observed that is perpendicular to the bundle and fully solvent-exposed. NMR experimental parameters confirm the existence of this short helix, which is proposed to serve as a recognition helix for apoLp-III binding to lipoprotein surfaces. The L. migratoria apoLp-III helix bundle displays several characteristic structural features that regulate the reversible lipoprotein binding activity of apoLp-III. The buried hydrophilic residues and exposed hydrophobic residues readily adjust the marginal stability of apoLp-III, facilitating the helix bundle opening. Specifically, upon lipoprotein binding the locations and orientations of the buried hydrophilic residues modulate the apoLp-III helix bundle to adopt a possible opening at the hinge that is opposite the recognition short helix, helix 4'. The backbone dynamics provide additional support to the recognition role of helix 4' and this preferred conformational adaptation of apoLp-III upon lipid binding. In this case, the lipid-bound open conformation contains two lobes linked by hinge loops. One lobe contains helices 2 and 3, and the other lobe contains helices 1, 4, and 5. This preferred bundle opening is different from the original proposal on the basis of the x-ray crystal structure of this protein (Breiter, D. R., Kanost, M. R., Benning, M. M., Wesenberg, G., Law, J. H., Wells, M. A., Rayment, I., and Holden, H. M. (1991) Biochemistry 30, 603-608), but it efficiently uses helix 4' as the recognition short helix. The buried interhelical H-bonds are found to be mainly located between the two lobes, potentially providing a specific driving force for the helix bundle recovery of apoLp-III from the lipid-bound open conformation. Finally, we compare the NMR structures of Manduca sexta apoLp-III and L. migratoria apoLp-III and present a united scheme for the structural basis of the reversible lipoprotein binding activity of apoLp-III.