Deposition Date
1995-06-20
Release Date
1995-10-15
Last Version Date
2024-02-14
Entry Detail
PDB ID:
1KRA
Keywords:
Title:
CRYSTAL STRUCTURE OF KLEBSIELLA AEROGENES UREASE, ITS APOENZYME AND TWO ACTIVE SITE MUTANTS
Biological Source:
Source Organism(s):
Klebsiella aerogenes (Taxon ID: 28451)
Method Details:
Experimental Method:
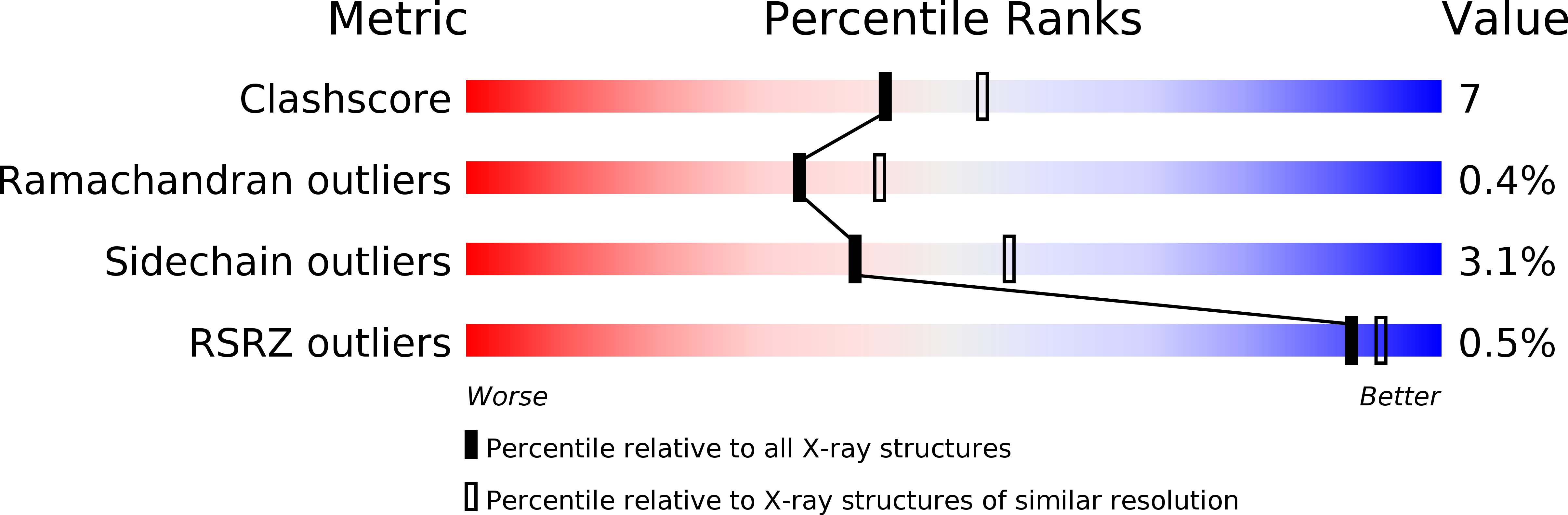
Resolution:
2.30 Å
R-Value Work:
0.19
R-Value Observed:
0.19
Space Group:
I 21 3