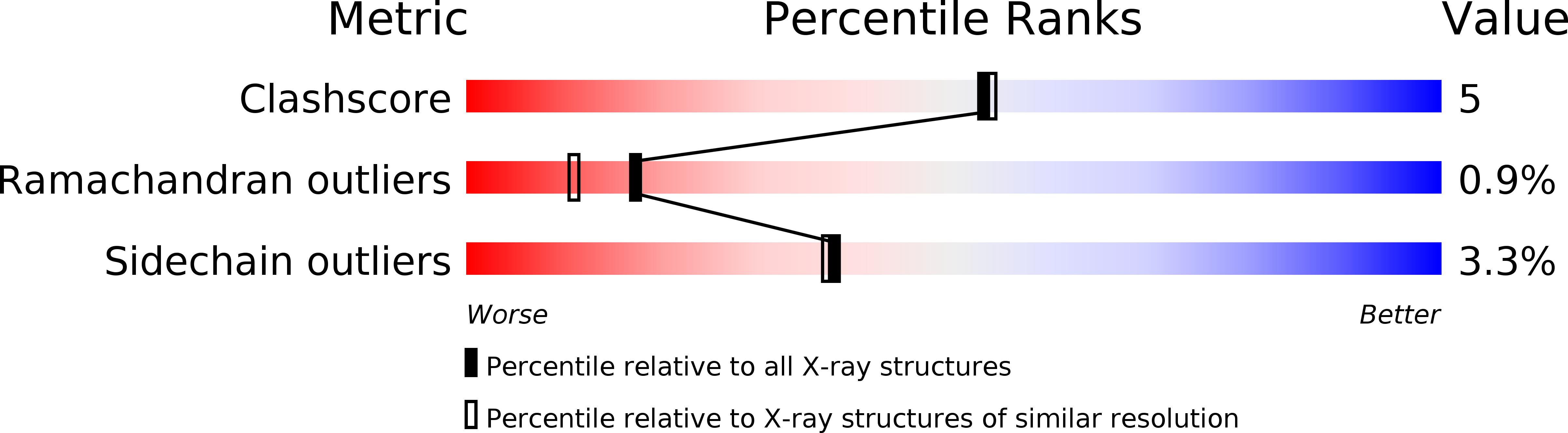Deposition Date
1996-07-30
Release Date
1997-01-27
Last Version Date
2024-10-16
Entry Detail
PDB ID:
1JPC
Keywords:
Title:
MANNOSE-SPECIFIC AGGLUTININ (LECTIN) FROM SNOWDROP (GALANTHUS NIVALIS) BULBS IN COMPLEX WITH MANNOSE-ALPHA1,6-(MANNOSE-ALPHA1,3)-MANNOSE-ALPHA1,6-(MANNOSE-ALPHA1,3)-MANNOSE
Biological Source:
Source Organism(s):
Galanthus nivalis (Taxon ID: 4670)
Method Details:
Experimental Method:
Resolution:
2.00 Å
R-Value Free:
0.22
R-Value Work:
0.18
R-Value Observed:
0.18
Space Group:
I 41 2 2