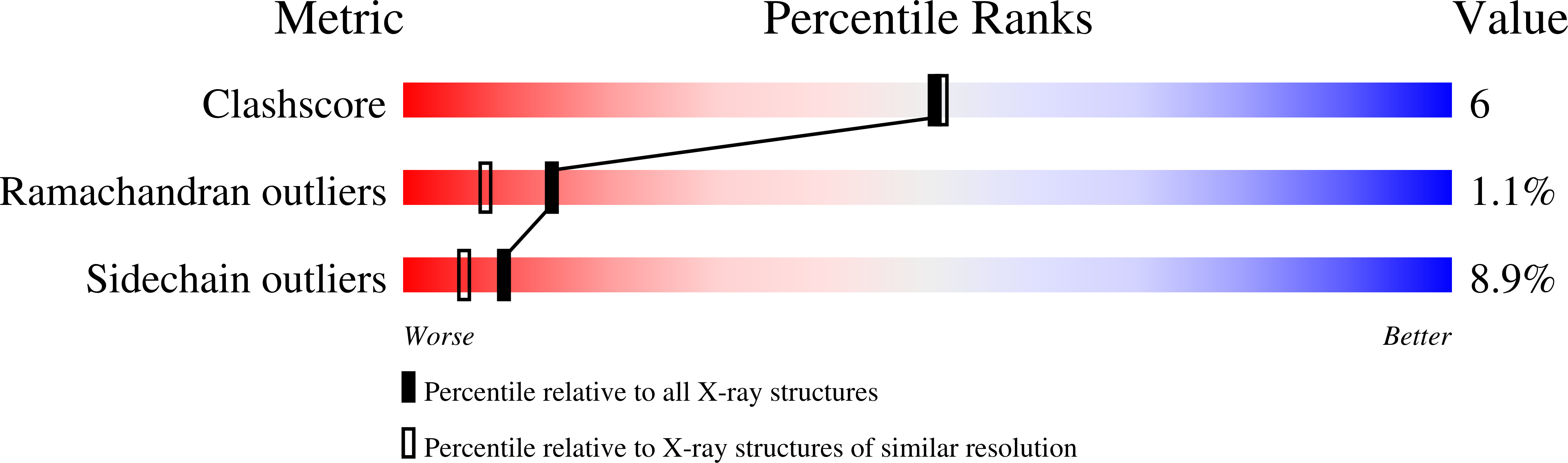
Deposition Date
2001-05-15
Release Date
2001-05-30
Last Version Date
2024-10-09
Entry Detail
PDB ID:
1J73
Keywords:
Title:
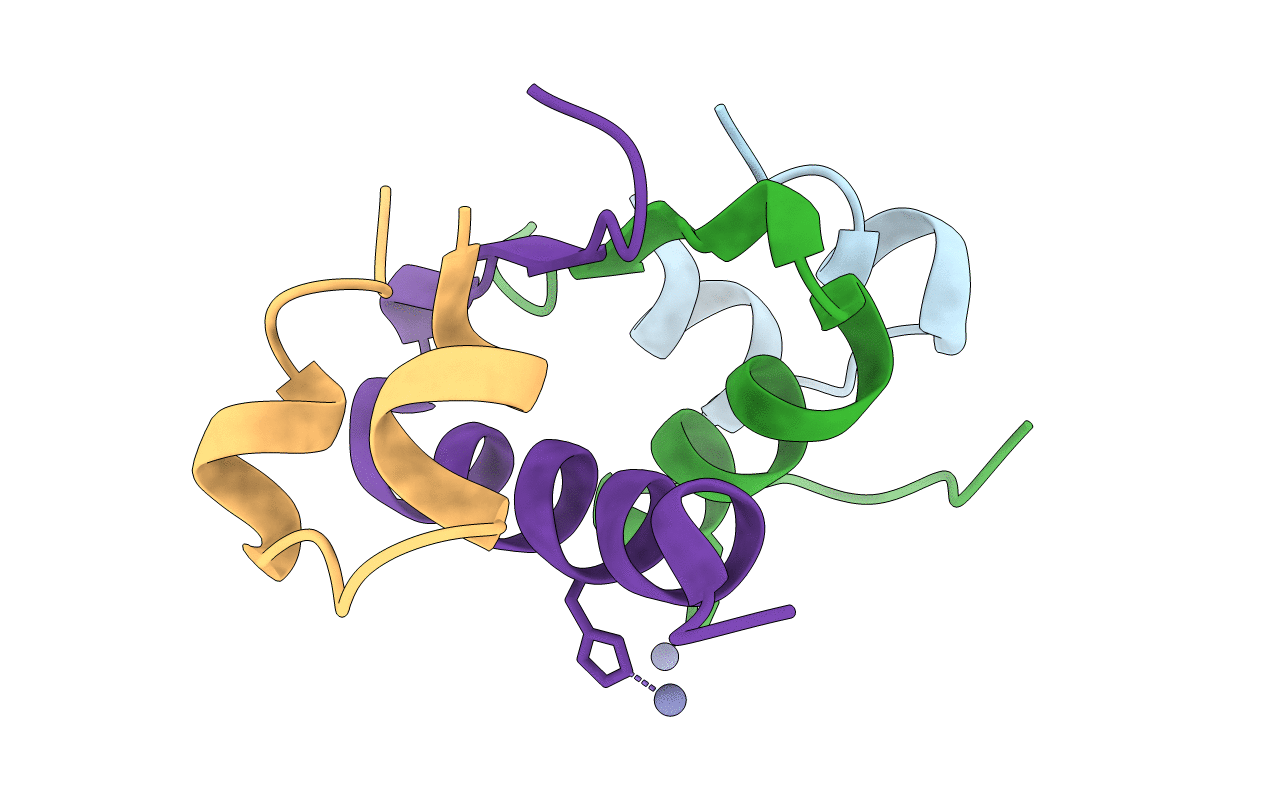
Crystal structure of an unstable insulin analog with native activity.
Method Details:
Experimental Method:
Resolution:
2.00 Å
R-Value Free:
0.27
R-Value Work:
0.20
Space Group:
H 3