Deposition Date
1994-06-22
Release Date
1994-10-15
Last Version Date
2024-02-07
Entry Detail
PDB ID:
1HBI
Keywords:
Title:
CRYSTAL STRUCTURE OF OXYGENATED SCAPHARCA DIMERIC HEMOGLOBIN AT 1.7 ANGSTROMS RESOLUTION
Biological Source:
Source Organism(s):
Scapharca inaequivalvis (Taxon ID: 6561)
Method Details:
Experimental Method:
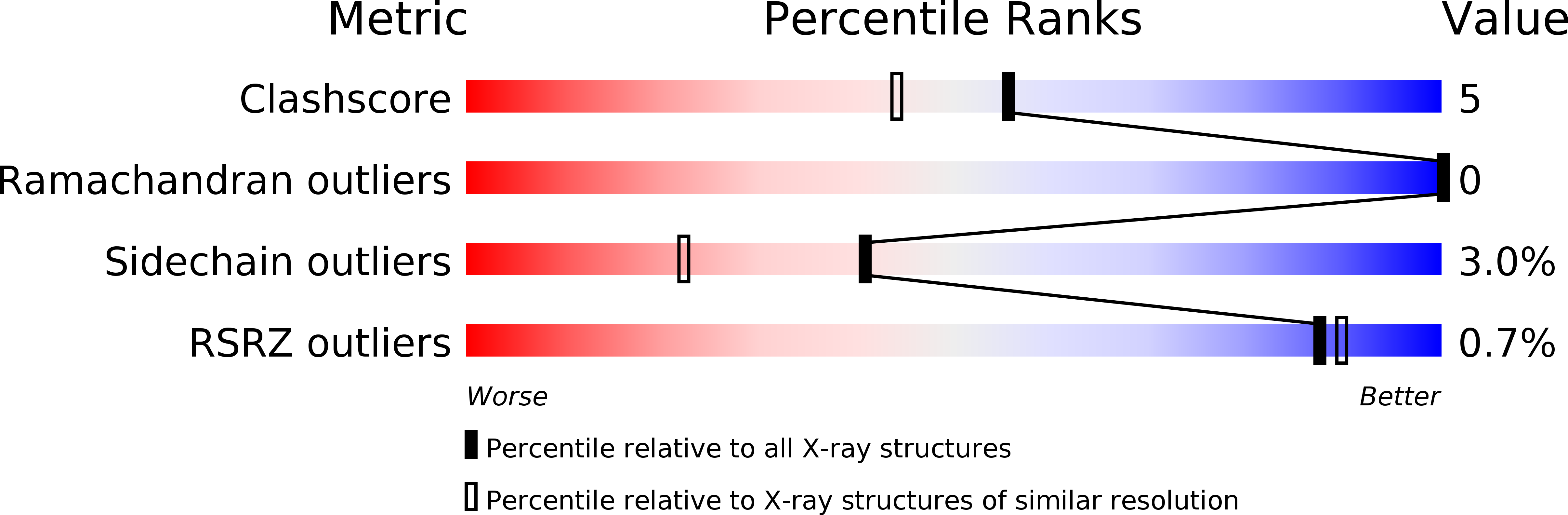
Resolution:
1.70 Å
R-Value Observed:
0.15
Space Group:
C 1 2 1