Deposition Date
2001-03-13
Release Date
2001-05-11
Last Version Date
2023-12-13
Entry Detail
PDB ID:
1H9M
Keywords:
Title:
Two crystal structures of the cytoplasmic molybdate-binding protein ModG suggest a novel cooperative binding mechanism and provide insights into ligand-binding specificity. PEG-grown form with molybdate bound
Biological Source:
Source Organism(s):
AZOTOBACTER VINELANDII (Taxon ID: 354)
Expression System(s):
Method Details:
Experimental Method:
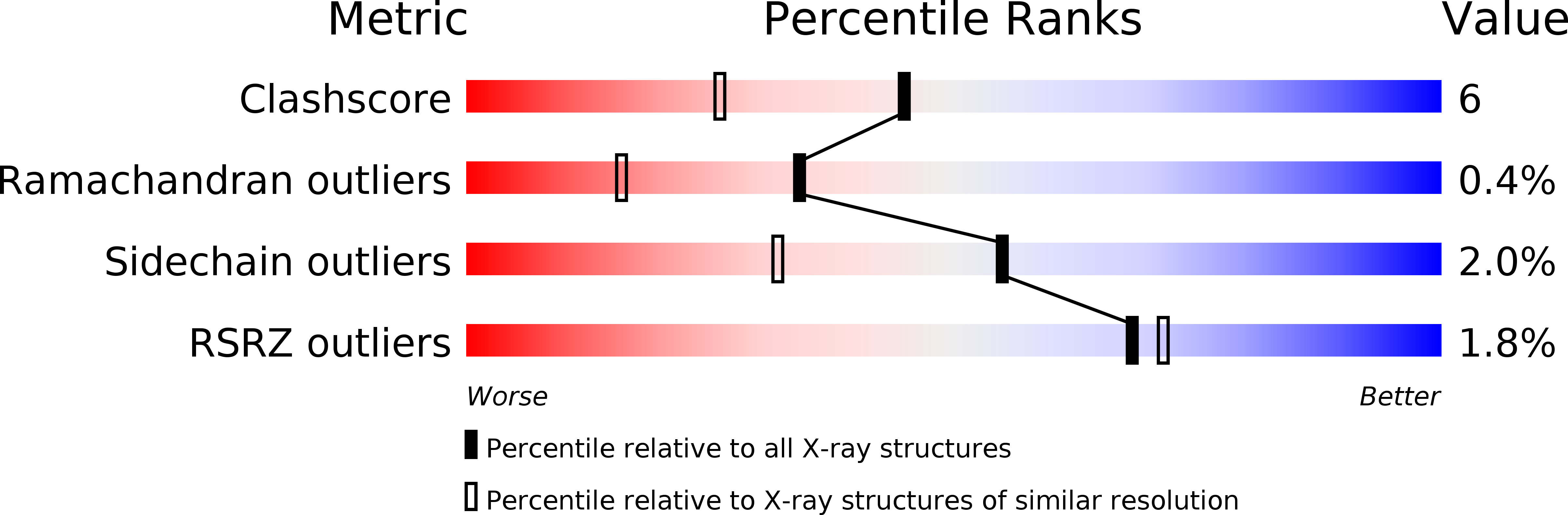
Resolution:
1.65 Å
R-Value Free:
0.22
R-Value Work:
0.19
Space Group:
H 3