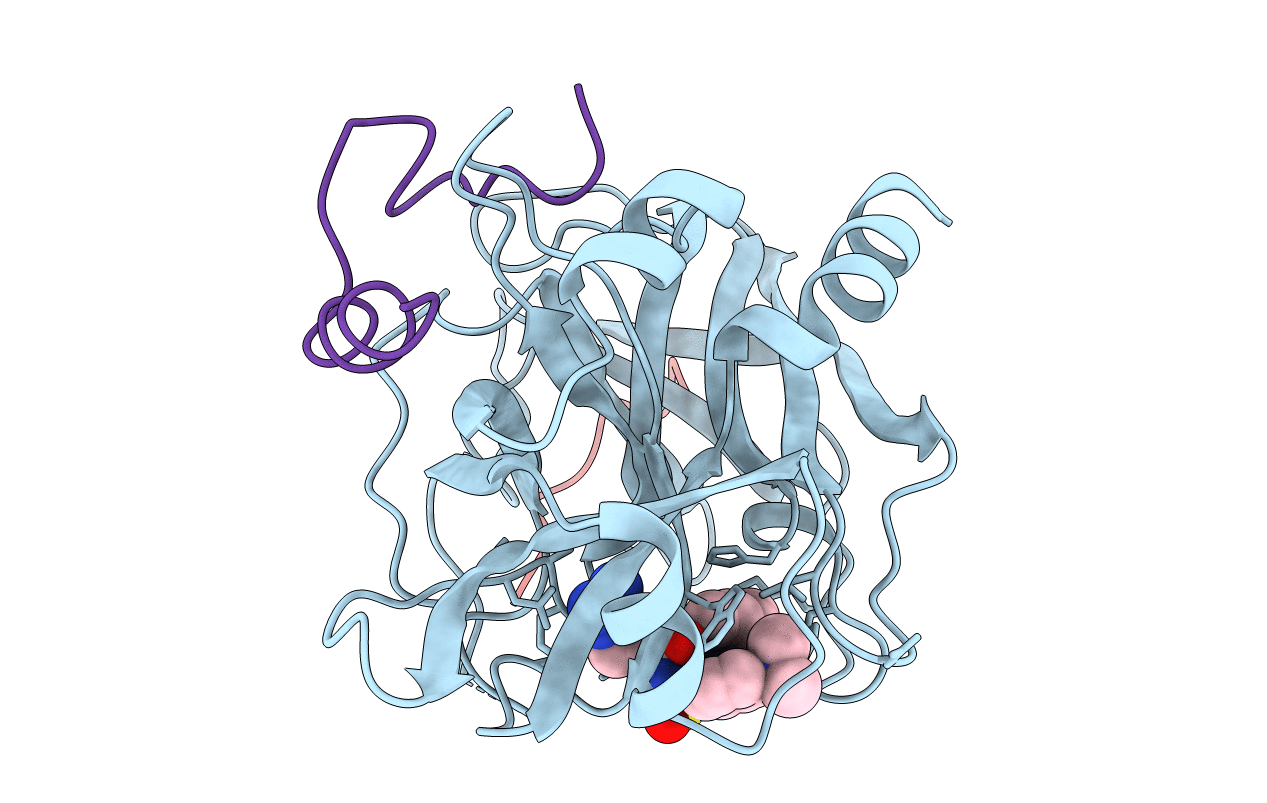
Deposition Date
1994-10-16
Release Date
1995-02-27
Last Version Date
2024-10-30
Entry Detail
PDB ID:
1FPC
Keywords:
Title:
ACTIVE SITE MIMETIC INHIBITION OF THROMBIN
Biological Source:
Source Organism(s):
Hirudo medicinalis (Taxon ID: 6421)
Homo sapiens (Taxon ID: 9606)
Homo sapiens (Taxon ID: 9606)
Method Details:
Experimental Method:
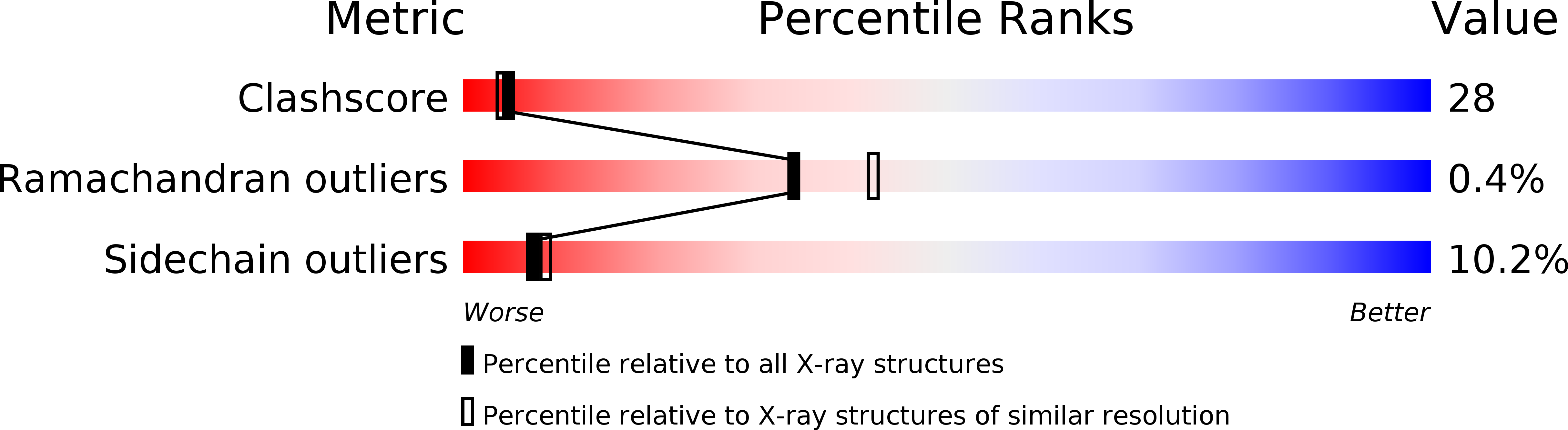
Resolution:
2.30 Å
R-Value Observed:
0.14
Space Group:
C 1 2 1