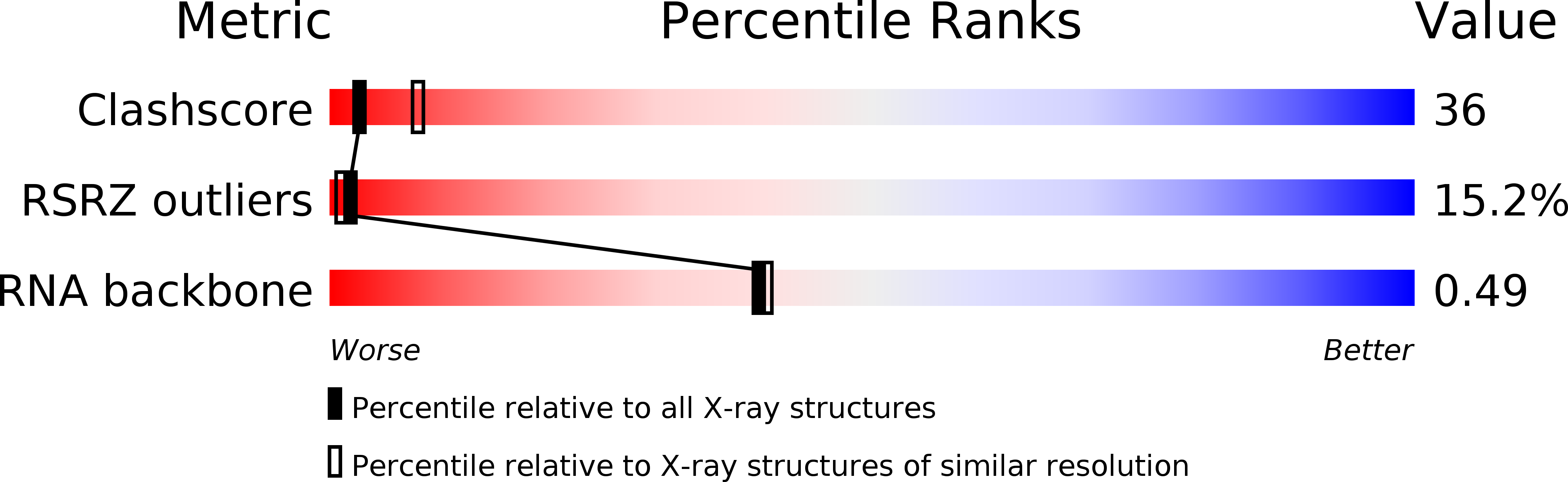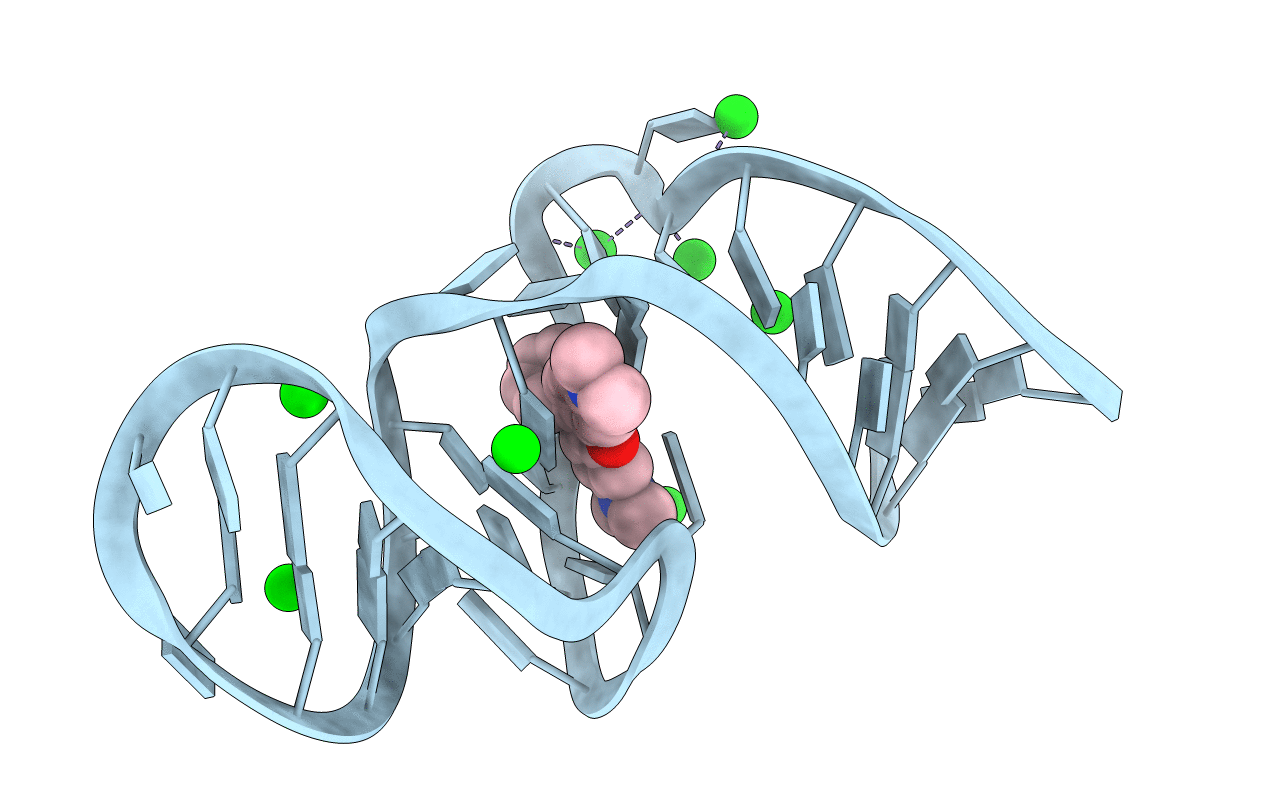
Deposition Date
2000-05-19
Release Date
2000-09-04
Last Version Date
2024-02-07
Entry Detail
PDB ID:
1F1T
Keywords:
Title:
CRYSTAL STRUCTURE OF THE MALACHITE GREEN APTAMER COMPLEXED WITH TETRAMETHYL-ROSAMINE
Method Details:
Experimental Method:
Resolution:
2.80 Å
R-Value Free:
0.28
R-Value Work:
0.26
R-Value Observed:
0.26
Space Group:
P 65 2 2