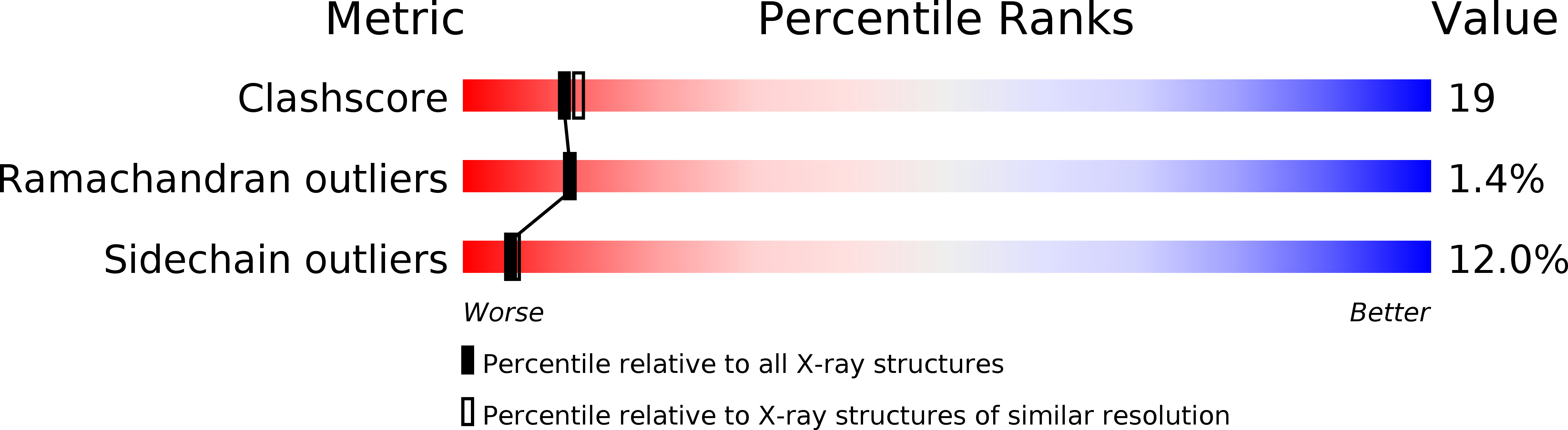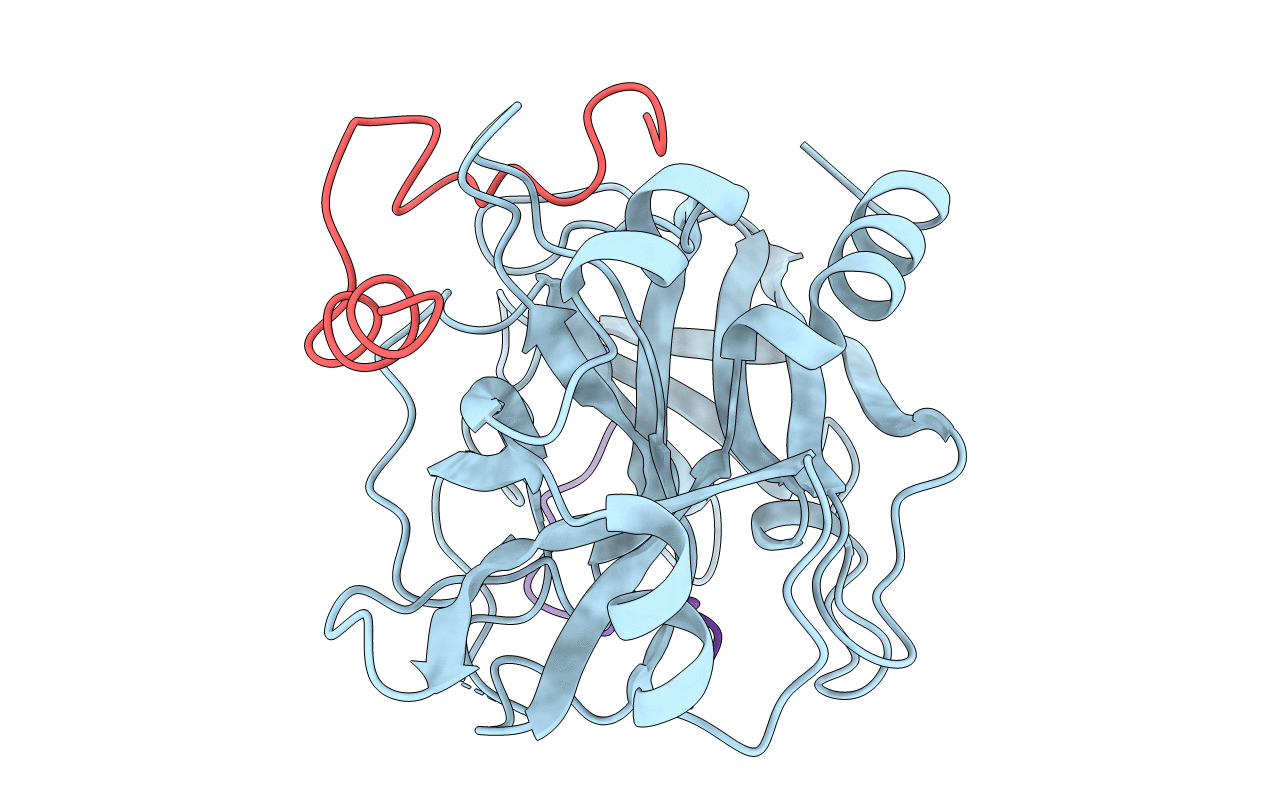
Deposition Date
1995-07-20
Release Date
1996-06-10
Last Version Date
2023-11-15
Entry Detail
PDB ID:
1DIT
Keywords:
Title:
COMPLEX OF A DIVALENT INHIBITOR WITH THROMBIN
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
Method Details: