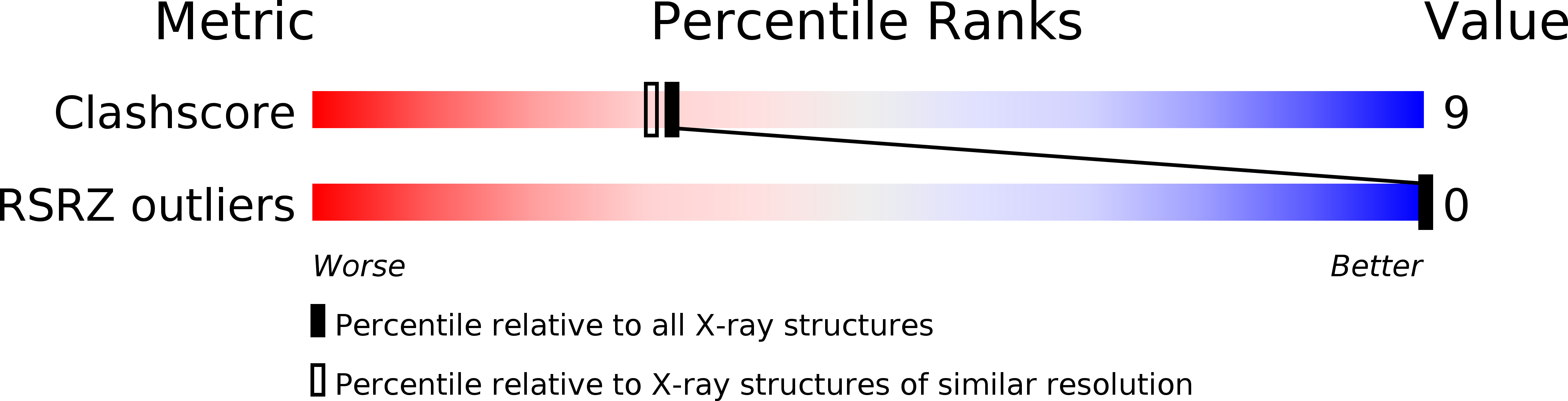Deposition Date
1992-03-02
Release Date
1993-01-15
Last Version Date
2024-02-07
Entry Detail
PDB ID:
1D63
Keywords:
Title:
CRYSTAL STRUCTURE OF A BERENIL-D(CGCAAATTTGCG) COMPLEX; AN EXAMPLE OF DRUG-DNA RECOGNITION BASED ON SEQUENCE-DEPENDENT STRUCTURAL FEATURES
Method Details:
Experimental Method:
Resolution:
2.00 Å
R-Value Observed:
0.18
Space Group:
P 21 21 21