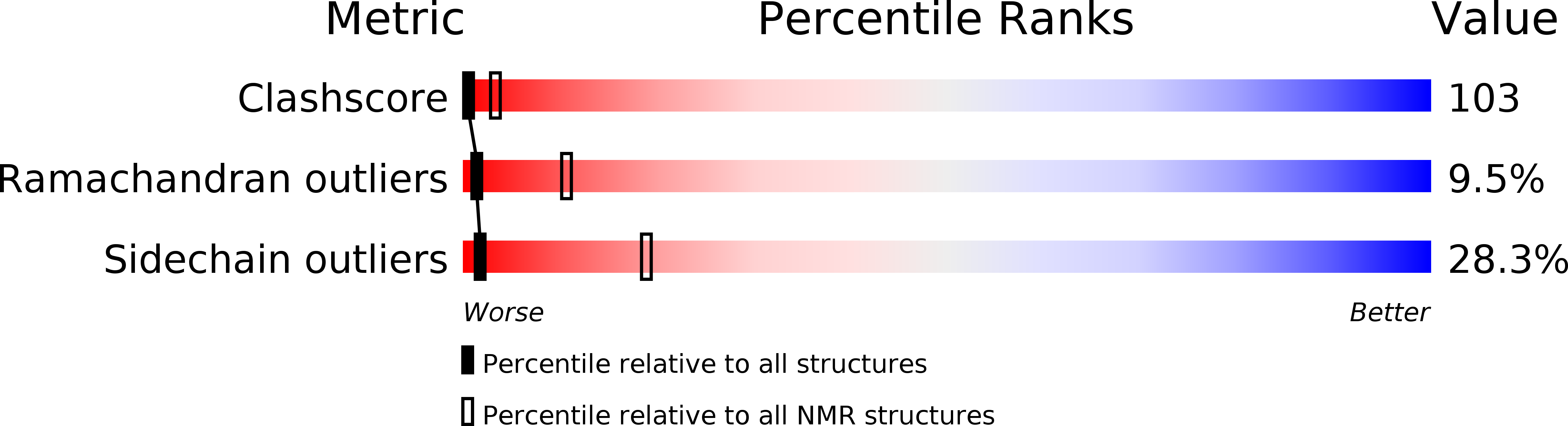Deposition Date
1995-10-03
Release Date
1996-11-08
Last Version Date
2024-11-13
Entry Detail
PDB ID:
1CSY
Keywords:
Title:
SYK TYROSINE KINASE C-TERMINAL SH2 DOMAIN COMPLEXED WITH A PHOSPHOPEPTIDEFROM THE GAMMA CHAIN OF THE HIGH AFFINITY IMMUNOGLOBIN G RECEPTOR, NMR
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
Expression System(s):
Method Details:
Experimental Method:
Conformers Submitted:
20