Deposition Date
1997-10-01
Release Date
1998-01-14
Last Version Date
2024-02-07
Entry Detail
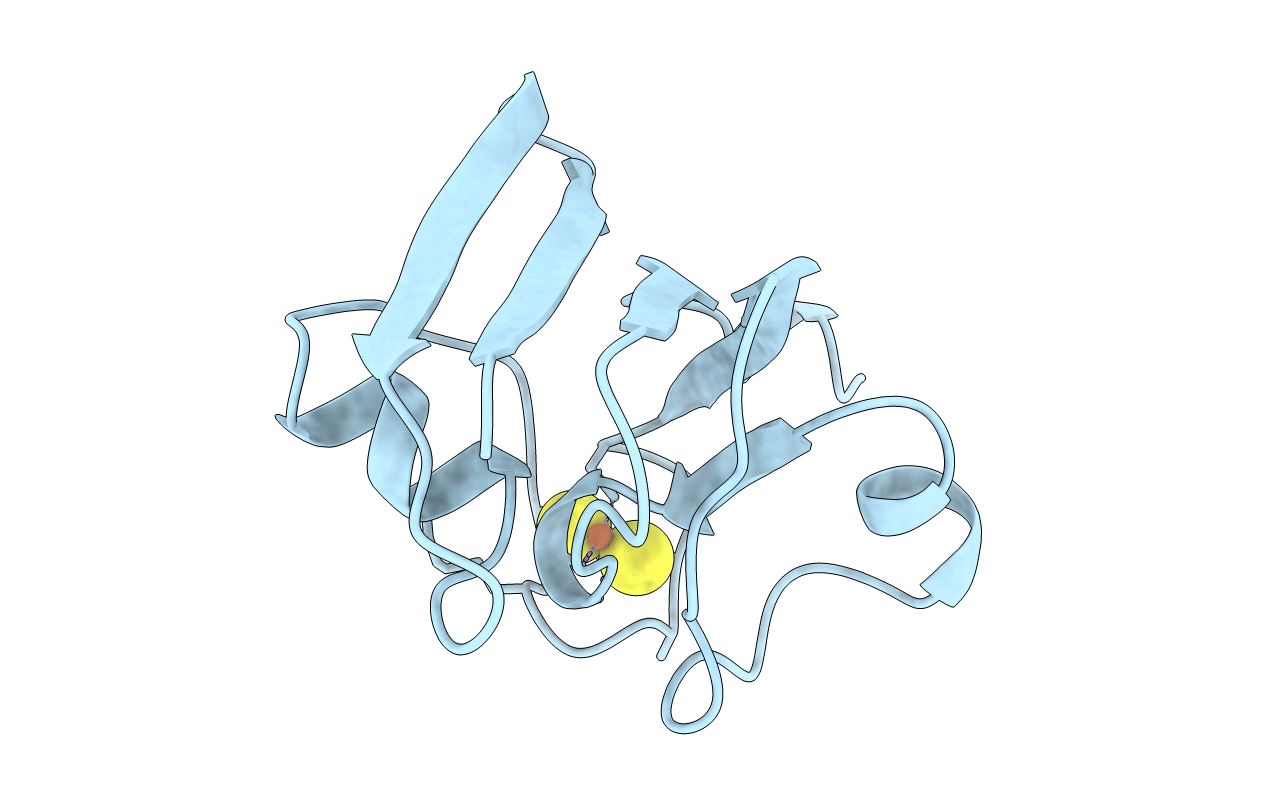
PDB ID:
1AWD
Keywords:
Title:
FERREDOXIN [2FE-2S] OXIDIZED FORM FROM CHLORELLA FUSCA
Biological Source:
Source Organism(s):
'Chlorella' fusca (Taxon ID: 3073)
Method Details:
Experimental Method:
Resolution:
1.40 Å
R-Value Free:
0.18
R-Value Observed:
0.14
Space Group:
I 4 3 2


