Deposition Date
1998-02-23
Release Date
1999-08-23
Last Version Date
2024-05-22
Entry Detail
PDB ID:
1A6B
Keywords:
Title:
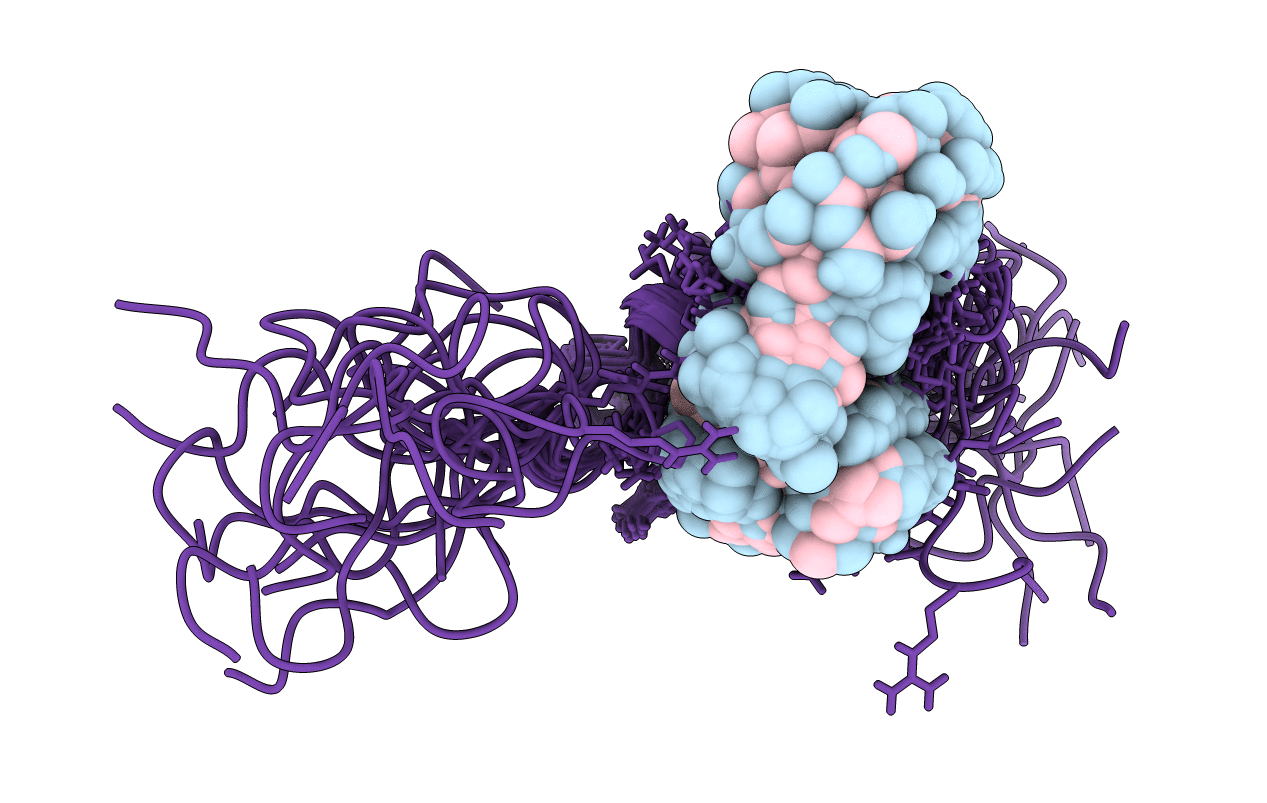
NMR STRUCTURE OF THE COMPLEX BETWEEN THE ZINC FINGER PROTEIN NCP10 OF MOLONEY MURINE LEUKEMIA VIRUS AND A SEQUENCE OF THE PSI-PACKAGING DOMAIN OF HIV-1, 20 STRUCTURES
Method Details:
Experimental Method:
Conformers Calculated:
50
Conformers Submitted:
20
Selection Criteria:
LEAST RESTRAINT VIOLATIONS, LOWEST TOTAL ENERGY