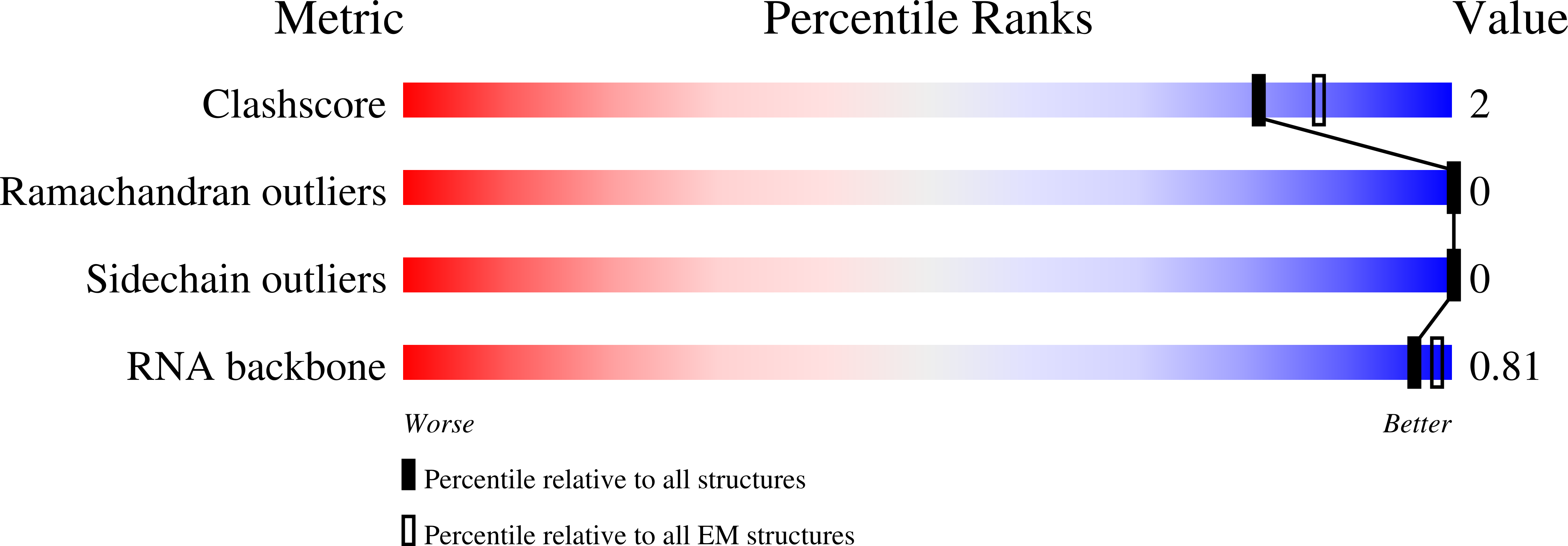
Deposition Date
2025-08-14
Release Date
2025-09-24
Last Version Date
2025-10-15
Entry Detail
PDB ID:
9Q1J
Keywords:
Title:
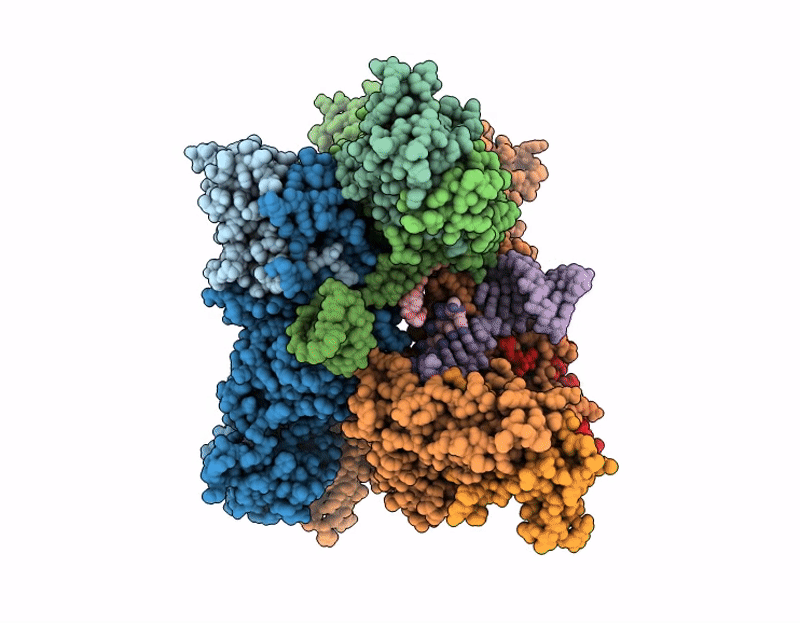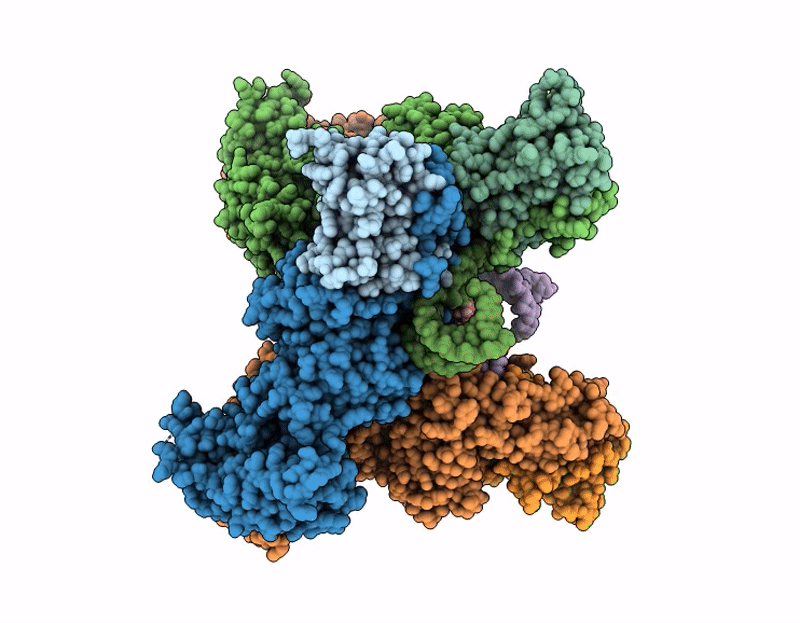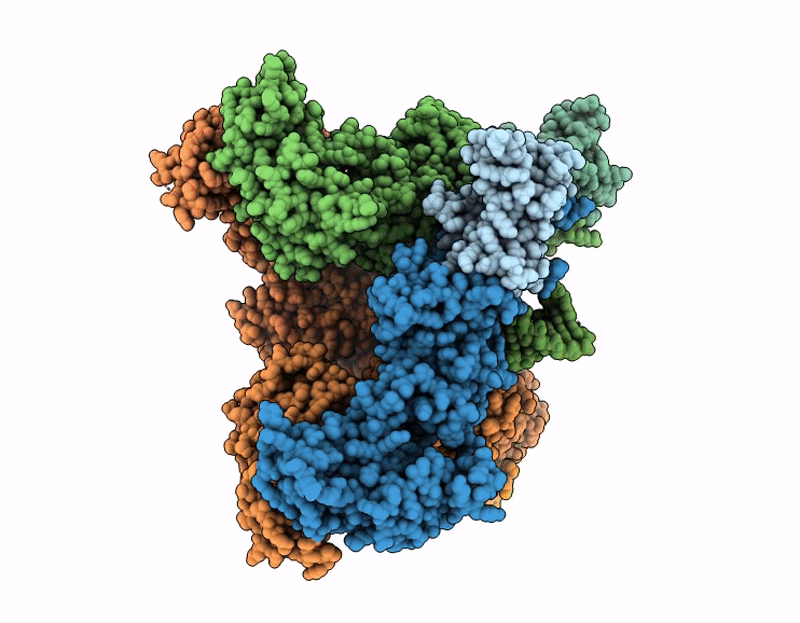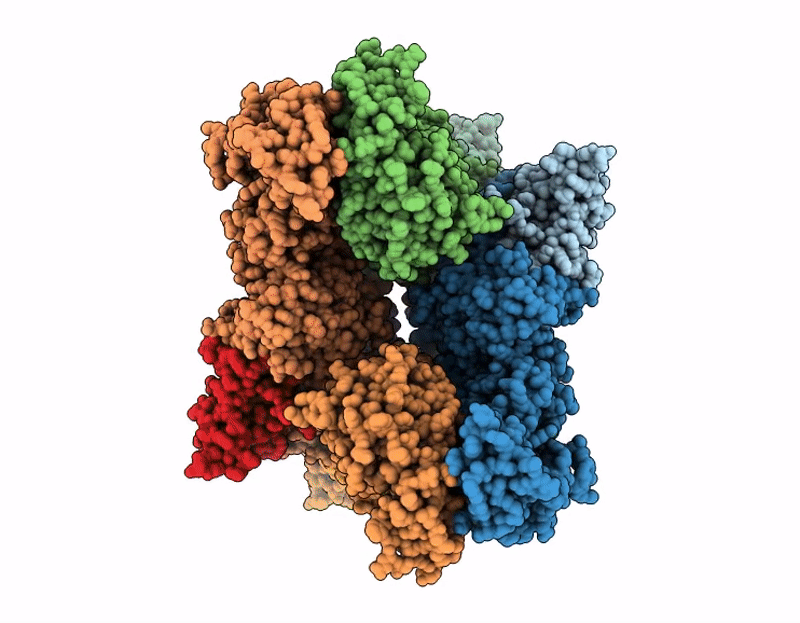
Cryo-EM structure of SARS-CoV-2 nsp10-nsp14 E191A mutant-T20P14-R complex
Biological Source:
Source Organism:
Host Organism:
Method Details:
Experimental Method:
Resolution:
2.81 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE