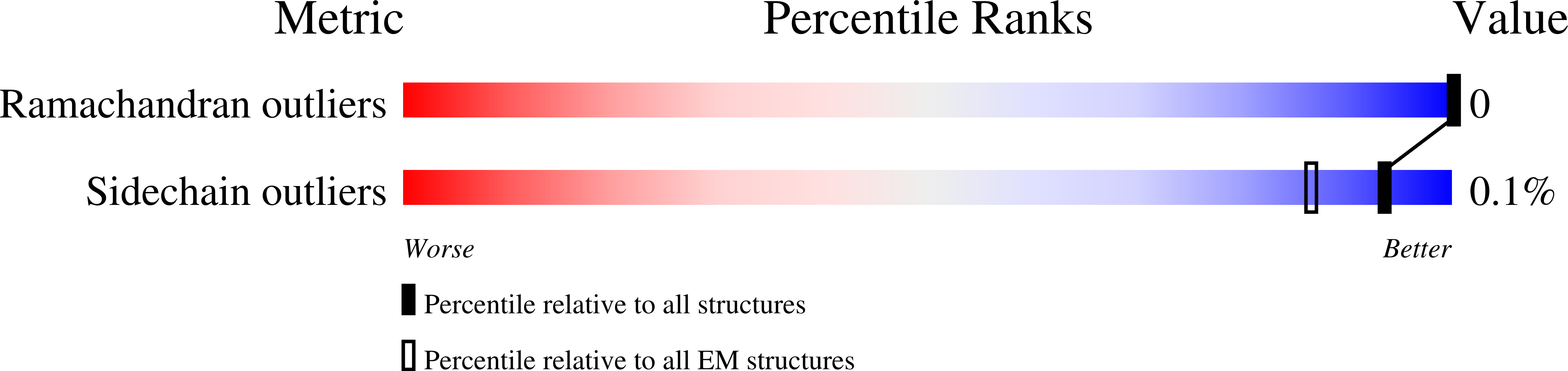
Deposition Date
2020-07-26
Release Date
2020-08-26
Last Version Date
2024-11-13
Entry Detail
PDB ID:
7JJI
Keywords:
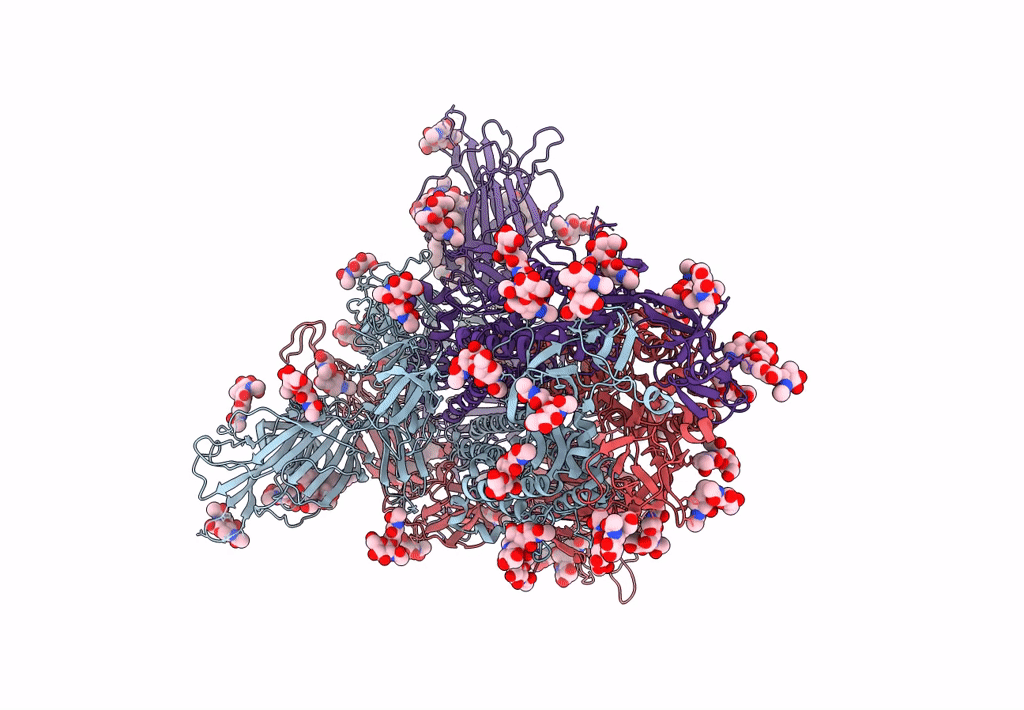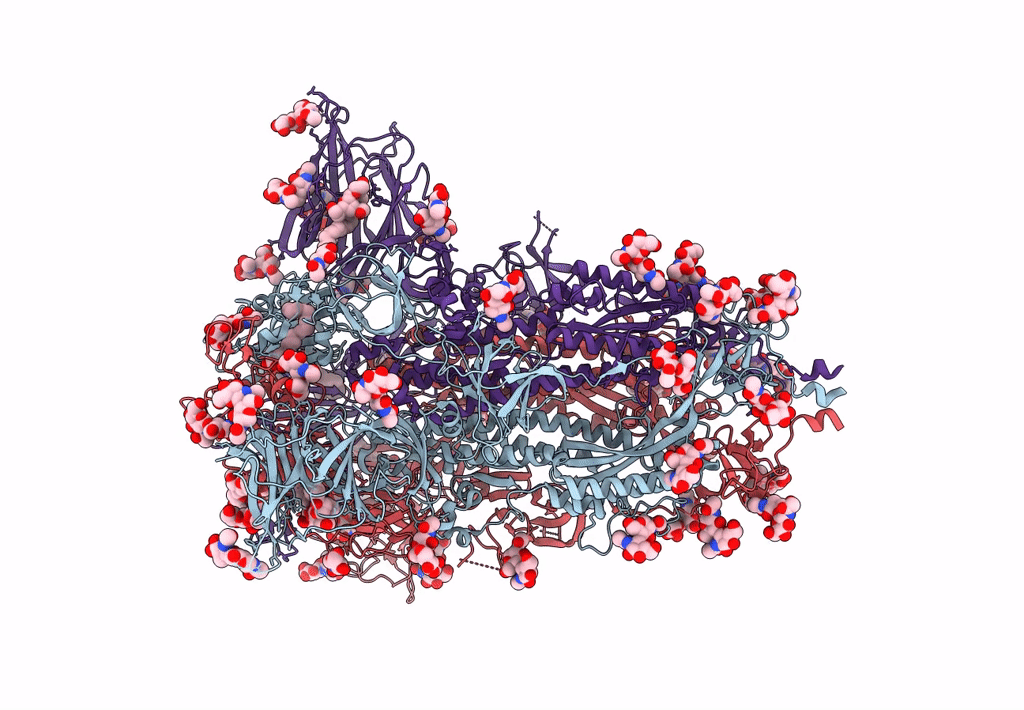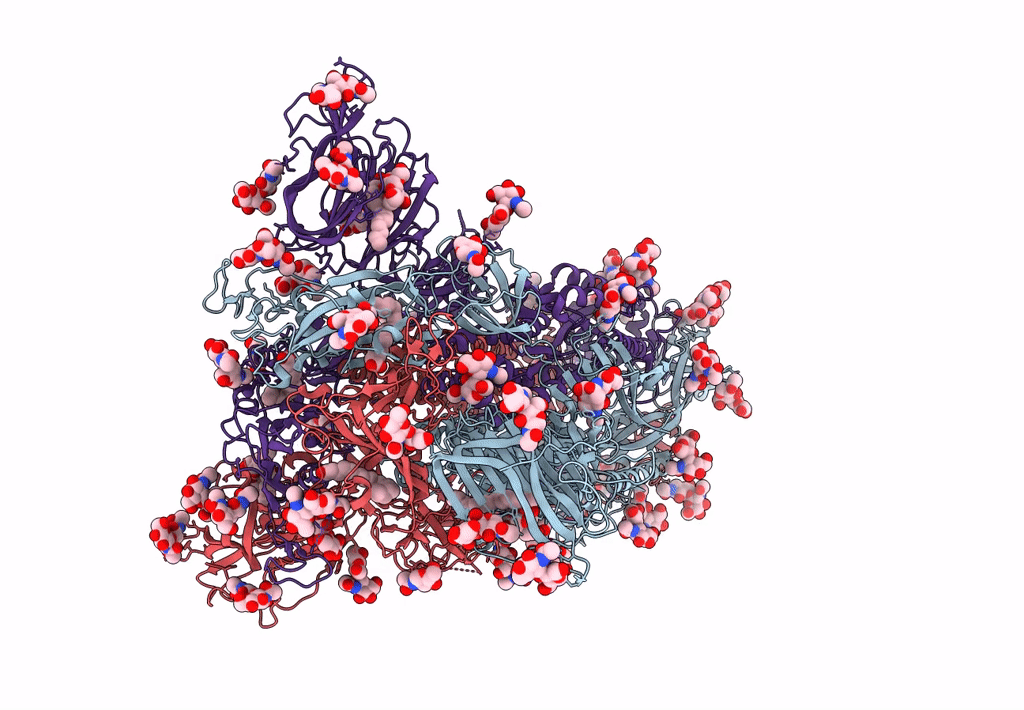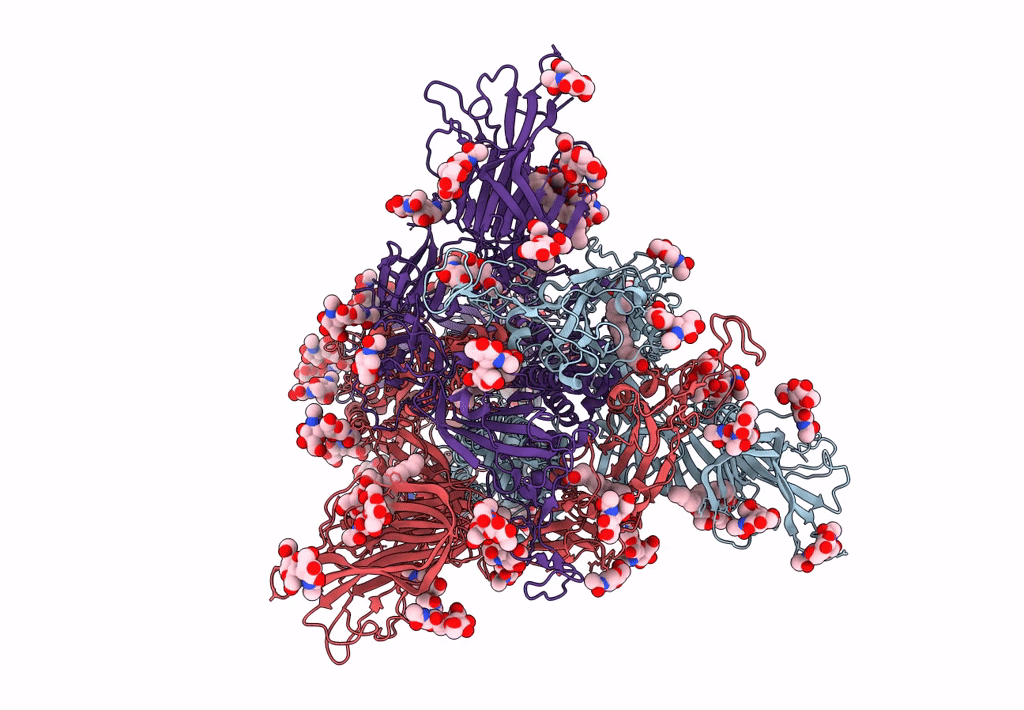
Title:
Structure of SARS-CoV-2 3Q-2P full-length prefusion spike trimer (C3 symmetry)
Biological Source:
Source Organism:
Host Organism:
Method Details:
Experimental Method:
Resolution:
3.60 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE